Amir Nadeem1,*, Sara Hayee2,*, Sadia Shakoor3, Naveed Akhtar4, Faheem Nawaz5, Farah Ashfaq2, Abdul Qayyum Khan Sulehria1,3
1Government Islamia Graduate College, Civil Lines, Lahore.
2Government Graduate College for Women, Samanabad, Lahore.
3Government College University, Lahore.
4Government Graduate College for Boys, Pattoki, Kasur.
5Government Khawaja Rafique Shaheed College for Boys, Lahore.
Received: 11-April-2023 / Revised and Accepted: 14-April-2023 / Published On-Line: 15-May-2023
ABSTRACT: This study intended to investigate the rotifer density and diversity from a flood plain. For this purpose, Balloki Headworks was selected. It is an area near the Ravi River that was affected by flooding. The sampling was done from April to June 2022. The highest diversity and density were observed in June and the lowest in April. Twelve species of rotifers were obtained from sampling, belonging to four different genera. Eight different physico-chemical parameters of water were measured using respective laboratory meters using standard guidelines. These parameters include the temperature of water and air, conductivity, dissolved oxygen, water pH, Oxygen saturation, salinity, total dissolved solids and turbidity. It was observed that Habrotrocha ligula (Bryce, 1913) and Rotaria rotatoria (Pallas, 1766) were the two most abundant species, while Philodina citrina (Ehrenberg, 1832) and Philodina acuticornis (Murray, 1902) were the least abundant.
Keywords: Planktonic rotifers, Flood plain, Habrotrocha, Rotaria, Philodina.
Introduction:
Extreme weather and climate changes bring about unusual rainy seasons, leading to flooding. Floods show spatio-temporal dimensions within limits of horizontal and vertical limits. Floods don’t show clear-cut boundaries, yet these are reservoirs marking flood plains and river channels [1]. Flood plains are aquatic, terrestrial transitional zones that are viewed as ecotones. These are formed at the boundaries of river systems [2]. Flood plains are considered resilient ecosystems [3]. These are also the most vulnerable landscapes of the world [4]. Very less water is available to sustain a floodplain landscape. The factors contributing to this situation include changes in the flow regime of dams, increased water extraction for human consumption etc. Flood plains and their associated rivers are hot spots of biodiversity and provide many ecosystems [5].
Zooplanktons, like rotifers, are key opportunist organisms that form the bases of food chains and the food web. Rotifers are small cosmopolitan pseudocoelomates belonging to the phylum Rotifera. They are found in various habitats involving lentic and lotic water bodies [6]. It has been described earlier that tropical and sub-tropical flood plains are the richest habitats for these delicate organisms [7]. Rotifers are important organisms that are a good source for larger zooplankton at higher trophic levels [8]. In aquatic ecosystems, rotifers are exposed to many changes involving physical, chemical and biological processes. These processes are linked to the physico-chemical factors of water bodies [9].
Because flood plains are not studied much for their biodiversity, the rotifers, which are a form of zooplankton that are quite small, were the primary subject of our investigation, which took place on a floodplain not far away. The effects of physico-chemical parameters on rotifer distribution in the said water body were also explored.
Materials and Methods:
The following materials and methods were used in the present study.
Study area:
A flood plain was selected as a study area near Balloki Headworks to study rotifers’ biodiversity. The latitude of Balloki Headworks is 31.22 (31°13″ 10N), and the longitude is 73.86 (73°51″ 35E). The area is almost 65 km away from Lahore. This floodplain is located on the southwest side, close to Phool Nagar, and is accessible via Multan Road. Sampling was done for three months, from April to June 2022.
Fig 1: Map of the study area. Courtesy Google Maps.
Water and Rotifer Sampling:
Sampling was done from five different sites of the littoral zone of the flood plain between 10am to 1pm. Before sampling, sample bottles were prepared by soaking in a 2% solution of HCl. Distilled water was used to rinse the bottles thrice. After that, the bottles were dried completely. Before the samples were taken at the sites, the bottles were washed again with the freely available sample water to remove any remaining residue.
A standard plankton mesh of size 50 µm was used for rotifer collection. These samples were preserved in 50ml bottles in 4% formalin solution [10-11].
Physico-chemical Analysis:
In order to study the effects of the hydro-chemical condition of the flood plain, eight physico-chemical parameters were studied. Table 1 shows the parameters and their respective meters used for measurement [12].
Table 1: Measurement of physico-chemical parameters.
| Physico-chemical Parameter | Meter Used |
| Water and atmospheric temperature | Laboratory thermometer, Model HANNA HI-8053 |
| Dissolved oxygen (water)
And Oxygen Saturation |
Dissolved Oxygen meter, Model YSI Eco Sense DO 200 |
| The pH of sample | pH meter, Model YSI Eco Sense pH 100 |
| Water Conductivity | Conductivity meter, Model YSI Eco Sense EC 300 |
| Total dissolved solids (TDS) | Total Dissolved Solids meter, Model YSI Eco Sense EC 300 |
| Salinity | Model YSI Eco Sense EC 300 |
| Turbidity | Turbidity meter, Model HANNA, Model No. HI-93704 |
Identification of rotifer:
By observation of morphology and behavioural characteristics, rotifers were classified up to the species level [13-17].
Rotifer counting:
For counting different rotifer species, a hemocytometer chamber or cell was used with the 60-100X magnification of an OLYMPUS microscope. The hemocytometer and a cover slip were cleaned with 70% ethanol before use. A Cannon LAICA HC 50/50 camera equipped with 5.0 megapixels’ worth of capability was mounted atop the microscope, and it was used to take photographs of rotifers.
Analysis of Diversity Indices:
The following diversity indices were calculated to study rotifers:
Shannon-Weaver index:
The Shannon-Weaver diversity index, is used to describe a community’s species diversity. This index considers species density and distribution. The Shannon-Weaver index values (H) can range from 0 to 4.6 using natural log (In).
The Shannon-Weaver equation calculated the index. It is given below:
H= Pi (InPi)
While:
Pi = The amount of each species that can be discovered in a certain sample.
Pi = ni / N [18]
While:
Ni = The total number of members of a certain species
N = Amount of each species’ representatives counted in the given sample
Simpson’s Index:
The Simpson’s Index of Dominance is a statistic that determines the likelihood that two individuals that have been arbitrarily selected from a population are of the same species. Its value can be anywhere between 0 and 1.
The value of Simpson’s Index of Dominance (D) was obtained with the help of Simpson’s equation. It is given below:
![]()
While:
D = Simpson’s Index of Dominance
N = All-species population in numbers
n = Per-sample species count
Sum of all
Simpson’s Diversity Index:
Simpson’s Diversity index (SDI) ranges from 0 to 1 and This index’s final value was calculated using the following formula:
SDI = 1-D
Species Richness:
Species Richness (SR) gives the number of species isolated from an area. The values for species richness were obtained by using Margalef’s Equation [19]:
SR = (S-1) / log N
While:
S = Species in total
N = No. of total individuals present in given sample
Species Evenness:
The values of species evenness were obtained by the equation given below [20],
E = H/ logn S
While:
S = Total No. of species
H = Shannon-Weaver Diversity Index
Statistical Analysis:
ANOVA or Analysis of Variance was applied to study the statistical differences. It was performed with the help of Minitab 13 software. All charts and graphs were created using Microsoft Excel.
Results:
The following results were obtained during the targeted time period of the present study.
Rotifer Density and Diversity:
Twelve rotifer species have been isolated from water samples of Balloki Headworks, which belonged to four different genera. Habrotrocha ligula (3.33) and Rotaria rotatoria (3.33) were the most abundant species. Philodina citrina (0.4) and Philodina acuticornis (0.46) were the two least abundant species. The most abundant genus was Philodina (41.17), and the least abundant genus was Adineta (11.61) (Table 2 & 3, Figure 2).
Table 2: Density and diversity of rotifer species (Ind/ml) isolated from Balloki Headworks (April-June 2022).
| Names of Rotifer Species | April
|
May
|
June
|
Mean |
| Adineta vaga (Davis, 1873) | 2.4 | 3 | 2.2 | 2.53 |
| Habrotrocha constricta (Dujardin, 1841) | 3 | 1.2 | 3.4 | 2.53 |
| Habrotrocha ligula (Bryce, 1913) | 3.4 | 3.6 | 3 | 3.33 |
| Philodina acuticornis (Murray, 1902) | 0.4 | 0 | 1 | 0.46 |
| Philodina citrina (Ehrenberg, 1832) | 0 | 0.4 | 0.8 | 0.4 |
| Philodina megalocephala (Hauer, 1936) | 3.8 | 3 | 1.2 | 2.6 |
| Philodina megalotrocha (Ehrenberg, 1832) | 1.4 | 1.4 | 1.2 | 1.33 |
| Philodina roseola (Bryce, 1913) | 0.6 | 1.4 | 1.4 | 1.13 |
| Philodina vorax (Janson, 1893) | 2.6 | 2.8 | 2 | 2.46 |
| Rotaria macroceros (Gosse, 1851) | 0 | 0.8 | 1 | 0.6 |
| Rotaria macrura (Ehrenberg, 1832) | 0 | 1.2 | 0.4 | 1.33 |
| Rotaria rotatoria (Pallas, 1766) | 3.6 | 3 | 3.4 | 3.33 |
| Total | 21. 2 | 21.8 | 21 |
Table 3: Relative percentage of rotifer genera collected from Balloki Headworks (April to June 2022).
| Genera | No. of Species | April | May | June | Mean |
| Adineta | 1 | 11.32 | 13.76 | 9.73 | 11.61 |
| Philodina | 6 | 41.50 | 41.28 | 40.70 | 41.17 |
| Rotaria | 3 | 16.98 | 22.93 | 21.23 | 20.39 |
| Habrotrocha | 2 | 30.18 | 22.01 | 28.31 | 26.84 |
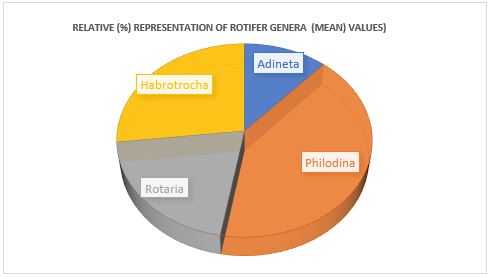
Fig 2. Percentage of each rotifer genus isolated from Balloki Headworks (April to June 2022).
Physico-chemical Parameters:
Physico-chemical parameters substantially impact the population dynamics of rotifers throughout the study period.
Water and Atmospheric Temperature (0C):
The present study’s water temperature ranged between 20.5 0C to 33 0C. The lowest atmospheric temperature recorded was 22.9 0C, while the highest was 35 0C (Table 4, Figure 3).
Dissolved oxygen (mg\L):
The mean DO values of rotifers ranged between 9.8 mg\L in April and 13 mg\L in June (Table 4, Figure 3).
pH:
Water pH ranged from 7.4 to 8.4 (Table 4, Figure 3).
Conductivity (µS/sec):
The conductivity of water was highest in June (362.8 µ/sec) and lowest in April (224.3 µ/sec) (Table 4, Figure 3).
Total Dissolved Solids (TDS) (mg/L):
Total dissolved solids were highest in June (0.1916 mg/L), while it was lowest (0.157 mg/L) in April (Table 4, Figure 3).
Salinity (ppt):
The highest value of 0.2 ppt of salinity was recorded in June and the lowest was documented in April and May with a value of 0.1 ppt (Table 4, Figure 3).
Oxygen Saturation (mg/L):
Oxygen saturation was maximum in April with a value of 150 mg/L; in June, it was 105 mg/L (Table 4, Figure 3).
Turbidity (FTU):
The highest mean value of turbidity, 25.94 FTU, was noted in April. As the turbidity increased, rotifers decreased in number. During June, the lowest mean value of 4.25 FTU was observed (Table 4, Figure 3).
Table 4: Values of Physico-chemical parameters (April to June 2022).
| Parameters | April | May | June | Mean |
| Water Temperature (0C) | 20.5 | 28.7 | 33 | 27.4 |
| Atmospheric temperature (0C) | 22.9 | 33.5 | 35 | 30.4 |
| Dissolved oxygen (mg\L) | 9.8 | 6.9 | 13 | 9.9 |
| pH | 7.4 | 8.4 | 7.84 | 7.88 |
| Conductivity (µS/sec) | 224.3 | 294 | 362.8 | 293.7 |
| Total Dissolved Solids (mg/L) | 0.157 | 0.182 | 0.1916 | 0.176 |
| Salinity (ppt) | 0.1 | 0.1 | 0.2 | 0.1 |
| Oxygen Saturation (mg/L) | 150 | 85 | 105 | 113.3 |
| Turbidity (FTU) | 25.94 | 19 | 4.25 | 16.39 |
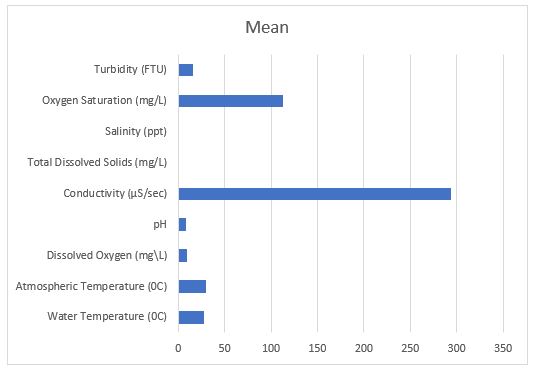
Fig 3. Values of Physico-chemical parameters (April to June 2022).
Diversity Indices:
The values of Shannon- Weaver Index showed a range from 2.0192 to 2.3318, the lowest in April and the highest in the month of June. The values of the Simpson Index of Dominance ranged between 0.1349 to 0.1076. The Simpson diversity Index showed value of 0.8650 in April and 0.8923 in June. Species evenness was nearly one in all months. Species richness was higher in June and lower in April (Table 5, Figure 4).
Table 5: Monthly variations in diversity, evenness and richness of species (April to June 2022).
| Month | Shannon-Weaver Index (H) | Simpson Dominance Index (D) | Simpson Diversity Index
(1-D) |
Species Richness
(SR) |
Species Evenness
(SE) |
| April | 2.01928 | 0.13494 | 0.86505 | 0.80307 | 0.91901 |
| May | 2.24629 | 0.11657 | 0.88342 | 1.00103 | 0.93677 |
| June | 2.33184 | 0.10768 | 0.89231 | 1.09718 | 0.93840 |
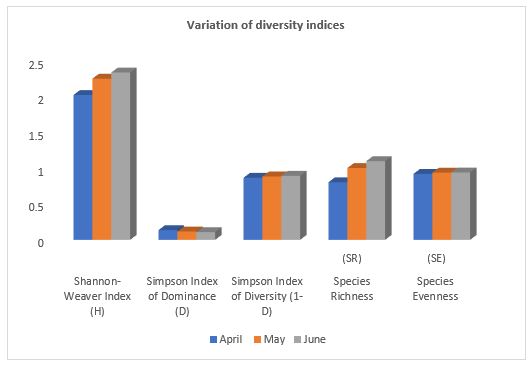
Fig. 4. Variation of Shannon-Weaver Index, Simpson Index of Dominance,
Simpson Index of diversity, Species Evenness and Richness during the study period (April to June 2022).
Analysis of Variance (ANOVA):
With the help of Analysis of variance (ANOVA), It was revealed that there was no significant difference (F=0.12, P=0.892) in the population density of rotifers among months. The maximum mean population was observed in May. It was lowest in June (Tables 2 & 6).
Table 6: Analysis of variance (ANOVA) between months and rotifers
(April to June 2022).
| Analysis of Variance for Rotifers | |||||
| Source | Degree of freedom = DF | Sum of squares = SS | Mean Square
= MS |
Distribution
= F |
Probability
= P |
| Months | 2 | 4.9 | 2.5 | 0.12 | 0.892 |
| Error | 12 | 256.8 | 21.4 | ||
| Total | 14 | 261.7 | |||
Discussion:
The present study revealed that the flood plain was a smaller water reservoir being a habitat with limitations supporting only 12 different rotifer species. There was an absence of key species of rotifer groups like Brachionus, Keratella etc. It proved that the species like Philodina, Adineta etc., are supported by moderate temperatures. The rotifer density was highest in May and depended on water temperature [21], [22]. It was observed that the two parameters dissolved Oxygen and Oxygen saturation, were higher in June, showing increased chances of ongoing chemical reactions in water. Usually, higher values of dissolved oxygen and oxygen saturation are related to higher rotifer density [11], but the present findings differ from the earlier studies. The pH ranged between 7.4 to 8.4. Usually, rotifers show a preference between 6.5 to 8.5. The recorded preference lies close to this range. Conductivity is a good indicator of the trophic state of a water reservoir. In the present study, the water conductivity gradually increased from April to June while rotifer density decreased. Total dissolved solids and salinity were maximum in June. Rotifers decreased with this increase. The greater the salinity, the lower will be the saturation point [11]. Turbidity was maximum in April 2022, and the Rotifer population got reduced with this increase in turbidity [23]. Our findings also relate to the above-discussed results. The values of diversity indices showed that this water body exhibited moderate rotifer diversity. The species evenness values showed the rotifers’ even distribution in the flood plain. It has been theorized that a more diverse array of species is responsible for a more balanced and uniform distribution of species and individuals within each species.
Author’s Contribution:
A.Q.K.S. conceived the idea and designed the research work; S.S., S.H., & A.N., performed the lab work, made all collections, acquired data. F.N., & F.A., executed data analysis and interpretation of data; S.H., & N.A., wrote the basic draft; A.N. did the language and grammatical edits or Critical revision. S.H., & A.N., did all the correspondence.
Funding: The publication of this article was funded by no one.
Author’s Conflict: Authors declare no conflict of interest.
Acknowledgement: The authors of this study are thankful to the Chairperson of the Zoology Department, Government College University, Lahore.
REFERENCES
[1] S. Eslamian and F. A. Eslamian, Flood Handbook: Principles and Applications, First. Boca Raton: CRC Press, 2022. doi: 10.1201/9781003262640.
[2] R. J. Naiman, H. Decamps, and M. E. McClain, Riparia: ecology, conservation, and management of streamside communities. Elsevier, 2010.
[3] M. C. Thoms, “Floodplain–river ecosystems: lateral connections and the implications of human interference,” Geomorphology, vol. 56, no. 3–4, pp. 335–349, Dec. 2003, doi: 10.1016/S0169-555X(03)00160-0.
[4] K. Tochner et al., Floodplains: Critically threatened ecosystems. Aquatic ecosystems, Trends and Global Prospects. Cambridge University Press, London, UK., 2008.
[5] C. Fischer et al., “The ‘Habitat Provision’ Index for Assessing Floodplain Biodiversity and Restoration Potential as an Ecosystem Service—Method and Application,” Front. Ecol. Evol., vol. 7, Dec. 2019, doi: 10.3389/fevo.2019.00483.
[6] A. Orstan, “Introduction to the Rotifera,” 1999, [Online]. Available: un.uobasrah.edu.iq/lectures/16394.pdf
[7] H. Segers, C. S. Nwadiaro, and H. J. Dumont, “Rotifera of some lakes in the floodplain of the River Niger (Imo State, Nigeria),” Hydrobiologia, vol. 250, no. 1, pp. 63–71, Jan. 1993, doi: 10.1007/BF00007495.
[8] J. A. Shah, A. K. Pandit, and G. M. Shah, “Rotifer community in relation to limnological characteristics of Wular lake in Kashmir Himalaya,” Ceylon J. Sci., vol. 46, no. 2, p. 49, Jun. 2017, doi: 10.4038/cjs.v46i2.7429.
[9] L. May and M. O’Hare, “Changes in rotifer species composition and abundance along a trophic gradient in Loch Lomond, Scotland, UK,” in Rotifera X, Berlin/Heidelberg: Springer-Verlag, pp. 397–404. doi: 10.1007/1-4020-4408-9_41.
[10] W. Koste, Rotatoria. Die Radertiere Mitteleuropas, Second. Gebruder Borntraeger, Berlin, Stuttgart: V. 1, text, 673 p.; V. 2, 234 plates, 1978.
[11] A. Q. K. Sulehria, R. Mushtaq, and M. Ejaz, “Abundance and composition of Rotifers in a pond near Balloki Headworks,” J. Anim. Plant Sci., vol. 22, no. 4, pp. 1065–1069, 2012.
[12] W. Federation and A. Association, Standard methods for the examination of water and wastewater, 21st ed. (American Public Health Association(APHA)). Washington DC, USA, 2005.
[13] R. W. Pennak, Fresh-water invertebrates-of the United States, 2nd ed. John Wiley and Sons, New York, USA. p.803, 1978.
[14] W. T. Edmondson, H. B. Ward, and G. C. Whipple, Freshwater Biology, 2nd ed. John Wiley and Sons. New York and London., 1959.
[15] S. K. Battish, Freshwater zooplankton of India. Oxford & IBH Publishing Company, Janpath, New Delhi, India., 1992.
[16] H. Segers, “Annotated checklist of the rotifers (Phylum Rotifera), with notes on nomenclature, taxonomy and distribution,” Zootaxa, vol. 1564, no. 1, pp. 1–104, Aug. 2007, doi: 10.11646/zootaxa.1564.1.1.
[17] L. H. Hyman, The Invertebrates. Vol. III. Acanthocephala, Aschelminthes and Entroprocta. McGraw-Hill, New York. p.55, 1951.
[18] M. Omori and T. Ikeda, Methods in marine zooplankton ecology. Wiley Interscience, New York. USA, 1984.
[19] R. Margalef, “Diversidad de especies en las comunidales naturales,” Publ. Inst. Biol. Apl., vol. 9, no. 1, pp. 5-27 (In Spanish), 1951.
[20] E. C. Pielou, “The measurement of diversity in different types of biological collections,” J. Theor. Biol., vol. 13, pp. 131–144, Dec. 1966, doi: 10.1016/0022-5193(66)90013-0.
[21] Ahlstrom Elbert H., A quantitative study of Rotatoria in Terwilliger’s Pond, Put-in-Bay, Ohio. Ohio State University Press, Ohio, USA, 1934.
[22] W. C. Allee and G. M. Rosenthal, “Group Survival Value for Philodina Rosola, A Rotifer,” Ecology, vol. 30, no. 3, pp. 395–397, Jul. 1949, doi: 10.2307/1932623.
[23] H. Zimmermann, “The microbial community on aggregates in the Elbe Estuary, Germany,” Aquat. Microb. Ecol., vol. 13, pp. 37–46, 1997, doi: 10.3354/ame013037.