Brohi1, R. Ali Arain1, A. Jabbar Laghari1, L. Ali Zardari1,*
1Department of Chemistry, Shaheed Benazir Bhutto University, Shaheed Benazirabad, Sindh Pakistan
*Correspondence: drabduljabbar.laghari@sbbusba.edu.pk
PJEST. 2025, 6(1); https://doi.org/10.5281/zenodo.17775450 (registering DOI)
Received: 13-March-2025 / Revised and Accepted: 30-Nov-2025 / Published On-Line: 03-Dec-2025
ABSTRACT: Nanoparticles (NPs) have been an area of research for the last decade due to their exceptionally small size and expansive surface area, resulting in distinctive physical and chemical transformations compared to bulk materials. The application of nano-biotechnology in health and medicine necessitates a strong coordination among physical scientists, chemists, and biologists. The green method was utilized to synthesize stable silver nanoparticles (Ag-NPs) using Setaria verticillata (SV) and Azadirachta indica (AI) seed exudates as reducing and capping agents. UV-visible and FTIR spectrometry were used to characterize the AgNPs physicochemical properties. In vitro toxicity testing of Ag-NPs produced from SV on adult earthworms (Lumbricina) showed substantial inhibition (P < 0.05). Antimicrobial efficacy of 0.72µg/mL Ag-NPs was tested against gram-positive (Bacillus amyloliquefaciens, Bacillus anthracis, S. warneri, and Staphylococcus aureus) and gram-negative bacteria (E. coli, Acinetobacter baumannii) suppressing pathogen proliferation. In contrast, the Ag-NPs from AI outperformed in killing all tested pathogens. Due to their thick peptidoglycan (PG) covering, Gram-positive bacteria are less vulnerable to Ag-NP penetration, hence their antibacterial activity was tested against several strains. This study examines how nano-catalysts degrade MB and CR. Eco-friendly NaBH4 usage. Additionally, kinetic models were used to evaluate adsorption kinetics. This research also highlights how the nanocatalyst affects NaBH4’s ecologically sustainable degradation of MB and CR. Additionally, Ag-NPs lowered CR and MB, suggesting their catalytic characteristics might eliminate commercial organic dyes. This research will help formulate eco-friendly bactericides and catalysts for biological applications.
Keywords: Green synthesis, Nanoparticles, Toxicity, Seed
- Introduction:
Nanoparticles (NPs) have been an area of research for the last decade owing to their exceptionally tiny size and expansive surface area, resulting in distinctive physical and chemical transformations compared to bulk materials [1]. The materials those having Structures at the atomic or molecular level with at least one dimension within the range of 1 to 100 nanometers are referred to as nanoparticles (NPs) [2]. The application of nano-biotechnology in health and medicine necessitates a strong coordination among physical scientists, chemists, and biologists [3]. The characteristics of NPs are contingent on their size and shape, leading to diverse usage ranging from bio-sensing, catalysts, and optics to bactericidal activity, transistors, memory schemes, electrometers, and sensors [4]. NPs are produced from different materials such as metal oxides, metals, silicates, biomolecules, and organic polymers. Moreover, they can be produced in various shapes i.e., spheres, platelets, cylinders, and tubes. Surface modification techniques are commonly applied to enhance the NPs properties for specific applications. Thus, due to extensive diversity in their shape, morphologies, particle medium and chemical composition makes NPs interesting subjects for novel research.
The production and application of NPs are enhanced due to their vast usage in many areas. The NPs can be classified into two main types i.e., organic NPs and inorganic NPs. Amongst them, noble-metal NPs such as silver (Ag) have gained significant attention from scientists due to their application in the bio-medical field, textile coatings, and cosmetics products [5], [6]. The usage of Ag-based NPs in many medical products increases due to their antimicrobial properties. The synthesis of Ag-NPs can be achieved through the physical (Laser ablation and evaporation-condensation processes), chemical (chemical reduction process), and biological approaches [7], [8]. Amongst them, biological techniques are cost-effective and environmentally friendly approaches due to the utilization of natural stabilizing and reducing substances. Moreover, the green synthesis of Ag-NPs from natural materials eliminates the need for toxic and harmful chemical usage.
Many researchers produced the Ag-NPs from the bacterial strain (Pseudomonas stutzeri AG259) and enzymes of fungi as fungi cells effective in capturing the Ag+ ions [9], [10]. The plant-based synthesis of Ag-NPs shows greater productivity because they possess greater metabolites involved in reduction as compared to the microbial process. The primary phytochemicals such as organic acids, quinones, and Flavones play vital role in significantly reducing Ag+ ions, thereby actively participating in the direct reduction and formation of Ag-NPs [11]. A few researchers investigated the different seed extracts such as Artocarpusheterophyllus lam, Melia azedarach, pomegranate, Linumusitatissimum L, Elettariacardamomom, and Fenugreek for the phyto-synthesis of Ag-NPs [12]–[14]. The release of free Ag+ from NPs causes the break of the cell wall of the bacteria, resulting in the death of the organism. Moreover, it is predicted that the 70-80 ug/g dietary intake of Ag is suggested to be non-toxic to the human body, thus it has been widely used in many commercial applications. In current research work, two types of seed extracts of plants Setariaverticillata (SV), and Azadirachtaindica (AI) are used to synthesize the Ag-NPs. These plants belong to Poaceae and Meliaceae family and grow in tropical regions of Asia. Moreover, they are widely used in many traditional medicine. The investigation of plant-based synthesis using seed extracts contributes to the understanding of phytochemicals’ role in reducing Ag+ ions. The antimicrobial properties of Ag-NPs and their potential in various commercial applications add practical significance to the research.
Nanoparticles (NPs) have been an area of research for the last decade owing to their exceptionally tiny size and expansive surface area. However, despite extensive progress, there is limited comparative work exploring how different plant seed extracts influence silver nanoparticle formation and their biomedical performance. This gap motivates the present study.
The aim of this work is to synthesize stable Ag-NPs using seed extracts and evaluate their properties. Specifically, this study focuses on (1) green synthesis of Ag-NPs using Setaria verticillata and Azadirachta indica seed extracts, (2) physicochemical characterization of the nanoparticles, and (3) assessment of their antimicrobial and catalytic performance..
- Materials and Methods:
2.1 Collection of plant material
Setariaverticillata (SV), seeds were collected from the locality of Nawabshah, Sindh Pakistan, as per guidelines of the Sindh University Department of Botany. In addition, A. Indica (AI) seeds were obtained from the local market of Karachi.

Fig 1: Setaria verticillata Plant and Azadirachta indica seeds
2.2 Synthesis of Ag-NPs
2.2.1 Synthesis of AgNO3 solution
To prepare 1mM AgNO3, 17mg of silver nitrate was weighed and poured into a beaker. Deionized (DI) water was added to achieve a total volume of 100 mL. This mixture serves as the silver salt precursor for NPs production.
2.2.2 Synthesis of SV and AI Seed Extract and Ag-NPs Production
After collection SV seeds were washed with DI water and soaked for one day. The resulting seed extract was bestrained and stored in the freezer until further experiment. Shared extraction steps are described once, followed by distinct parameters for the two seed types. Various concentrations of extract (10–40 percent) were used “based on preliminary tests showing that concentrations below 10 percent produced weak reduction activity, while concentrations above 40 percent caused rapid agglomeration and unstable colloids. For the synthesis of silver nanoparticles (Ag-NPs), various concentrations of SV extract (10-40 vol %) are mixed with a 1 mM AgNO3 solution. Similarly, the AI seeds were rinsed three times with DI water, and an extract was prepared by soaking 10 g of seeds in 100 mL DI water for one day with mild heating (60 °C). The seed extracts were filtered and stored in the freezer before the experiment. AgNO3 will serve as silver salt precursor for the reduction of Ag+ into Ag atoms. To achieve this reduction, several concentrations of AI seed extract, namely 10% (N1), 20% (N2), 30% (N3), and 40% v/v (N4) were mixed with a (1mM) aqueous AgNO3 solution at pH 8 for 30 minutes.
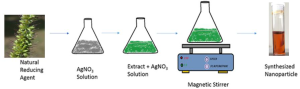
Fig 2: Graphical view of synthesis of Ag-NPs from Green Natural Material
2.3 Characterization of synthesized Ag-NPs
The produced Ag-NPs were characterized through UV–Vis and FTIR. “The EDX measurements were removed as the data were incomplete and not directly discussed in the results
2.4 Application of Produced Ag-NPs
In order to utilize the produced Ag-NPs in the medical field bacterial species were used in the experiment. Two different bacterial strains (B. anthracis, and E. coli) were procured from the Department of Zoology, University of Sindh Jamshoro. Moreover, the Lumbricina earthworm shall be collected from the river Indus. Earthworms were exposed for 48 hours under controlled temperature and moisture conditions, with untreated worms serving as the control group. Mortality, motility, and behavioral changes were recorded at 12-hour intervals following established protocols. The earlier analytical technique will be used to confirm the Lumbricina worm [15]. The Ag-NP bactericidal efficacy will be assessed against both Gram-negative strains (E. coli) and Gram-positive strains (B. anthracis).
The bacterial strains will be cultured on nutrient agar (HiMedia), and the disc diffusion method was used to determine the susceptibility of biologically synthesized AgNPs to the selected strains. In short, bacterial suspensions, each containing 1 x 107 CFU/mL, were evenly spread on nutrient agar plates using a glass spreader. Sterilized borer discs were then placed on the agar plates under aseptic conditions. The Ag-NPs solution was applied to each plate inoculated with the two different bacterial strains. After overnight incubation at 37 °C, the zones of inhibition were measured, and their lengths were calculated in millimeters using a meter ruler.
2.5 Catalysis
Double-distilled water was utilized to dilute a 1mM solution of methylene blue (MB) and congo red (CR) to achieve the optimal concentration of the dyes. A freshly prepared solution of sodium borohydride (NaBH4) was introduced into 3 mL of dye solution, followed by the addition of 400 µL of silver colloid [16]. Subsequently, the colloidal solution was agitated for a duration of five minutes. The observed decolorisation of the blend signifies the degradation of dyes. The reducing agent (NaBH4) discharges the color of methylene blue, indicating a decrease of MB to leucomethylene blue (LMB). The reaction without the nanocatalyst serves as a reference point. The absorption spectrum was observed at 2-minute intervals at a temperature of 25 °C.
- Results and Discussion
3.1 UV-Vis Spectroscopy of prepared Ag-NPs with SV and AI
Fig 3 illustrates the UV-visible spectra of SV and A. indica nanoparticles of silver generated with varying amounts of seed extract. A large absorption peak is seen between 350 and 550 nm after the reaction of AgNO3 with SV seed extract. Simultaneously, reducing the SV concentration from 40 vol% to 10 vol% resulted in a shift of absorption to longer wavelengths, followed by a notable drop in absorption intensity as shown in Fig 3a. Moreover, the surface plasmon resonance was detected at around 400 nm in both N1 and N2, regardless of the fact that N2 had a greater peak intensity than N1. Increasing A. indica seed extract concentrations (N3 & N4), however, led to a reduction in absorption intensity and wider peaks, which suggests particle agglomeration. Furthermore, as shown in Fig. 3b, inset, the color of the reaction mixture changed from light yellow to wine red with the addition of larger concentrations of 30% (N3) and 40% v/v (N4) of A. indica seed extract. In our instance, the A. indica seed extract quickly converted Ag+ into NPs, which is unique.
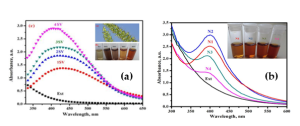
Fig 3: (a) UV-vis spectra for SV seed extract and varying quantities of extract from 10 vol% (1SV) to 40 vol% (4SV) in the synthesis of AgNPs; (b) UV–Vis spectra of biosynthesized Ag-NPs using A. indica seed extract with different concentrations (N1–N4). N1 = 10%, N2 = 20%, N3 = 30%, N4 = 40% v/v extract.
3.2 FTIR Analysis of AgNPs synthesis from Sv and AI seed
FTIR analyses were conducted to ascertain the different functional groups contained in the sample (Fig. 4.). The spectrum signals at 1621 and 1558 cm-1 relate to the existence of the aromatic –C-O vibrational mode, which is a distinctive peak for the amide band of carbonyl. Significantly, the bands 3271, 1560, 1387, 1041, and 613 cm-1 of the extract are displaced in the SV Ag-NPs. The absorption band at 3271 cm-1 in SV seed extract moved to a higher wavelength in nanoparticles of silver, whereas the 1621 cm-1 band shifted to a lower wavelength (1578 cm-1), perhaps indicating its role in the reduction of silver nitrate to silver nanoparticles [17].
The absorbance peaks of A. indica stabilized Ag-NPs (3273, 2924, 1576, 1457, 1039, and 517 cm−1) correspond primarily to flavones and terpenoids that are abundant in A. indica seed extract [18]. Peak shifts from 3244–3273 cm-1, 2928–2924 cm-1, 1663–1576 cm-1, 1507–1457 cm-1, 1022–1039 cm-1, and 518–517 cm-1 were observed in the FTIR spectral analysis of A. indica extract and Ag-NPs, as illustrated in Figure 4 b. The flavanones or terpenoids adsorbed on the Ag-NPs surface by interacting π-electrons or carbonyl groups [19]. This indicates the association of hydroxyl radical, carboxylate, and ketonic functional groups involved in forming and stabilize the Ag-NPs [20].
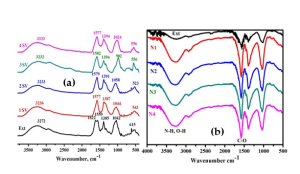
Fig 4: FTIR spectra of AgNPs at (a) varying SV seed extract concentrations from 10 vol% (1SV) to 40 vol% (4SV); (b) varying AI seed extract concentrations ranging from 10% to 40% v/v (N1–N4) combined with 1 mM silver nitrate
3.3 Antibacterial activity of Ag-NPs produced from SV and AI seeds
As can be seen in Figure 5, the good diffusion experiments demonstrated that the A. indica seed extract and the biosynthesized Ag-NPs were sensitive to the presence of potentially harmful bacteria. Significant zones of inhibition were found for N1, N2, and N3 against bacterial strains that ranged from 1.1 to 2.9 centimeters in length. On the other hand, a modest degree of inhibition was reported for N4 against both gram-positive and gram-negative bacterial strains (1.1 – 1.5 cm).
Due to the small size factor, the antimicrobial activity of N1 is higher than that of N2 and N3. Additionally, the presence of trace amounts of bio-moieties in N1 does not significantly contribute to the bactericidal activity of the compound since the bio-moieties may be derived from the extract [16]. The widths of the zones of inhibition in the instance of A. indica seed extract were found to fall somewhere in the range of 0.8 to 1.0 millimeters. The agglomeration of nanoparticles, which causes a reduced availability of nanoparticles to the membranes of the bacteria, is a plausible explanation for the decreased sensitivity of N4 against all of the bacterial species that were examined. This has previously been demonstrated by a number of studies [17], [18], [21], [22]. A greater concentration of seed extract results in an increase in the quantity of reducing agent. This leads to the simultaneous reduction of a large number of Ag+, which in turn leads to the development of more agglomeration, which in turn results in the production of larger nanoparticles.
The nanoparticles showed inhibition zones against all bacterial strains. The trend in inhibition correlates with nanoparticle size, where smaller and less agglomerated particles (especially from lower extract concentrations) achieved stronger interactions with bacterial Extract-derived phytochemicals also influenced stability, contributing to variability between SV and AI seed–based Ag-NPs
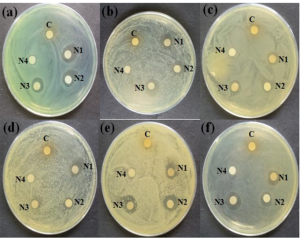
Fig 5: Efficacy of A. indica seed extract and AgNPs. (a) Bacillus anthracis, (b) Bacillus amyloliquefaciens, (c) Staphylococcus aureus, (d) Staphylococcus warneri, (e) Escherichia coli, and (f) Acinetobacter baumannii
In the case of SV seed extract AgNPs the well diffusion technique was used to assess antibacterial properties. Higher activity was reported against strains a, b, e, and f, whereas strains c and d showed modest inhibition at 0.75 μg.mL-1. The inhibitory zone in Fig.6 grew with higher seed extract concentrations used for NPs production. The 4SV sample showed the best antibacterial activity due to the shape of silver nanoparticles. Nanoparticles of tiny size have great bactericidal potential and high binding capacity [23], [24]. However, bio-synthesized silver nanoparticles show greater bactericidal efficacy against gram-negative pathogens (E. coli and Acinetobacter baumannii). We hypothesize that biogenic NPs interact with bacterial strains, causing cell wall rupture and ROS production, leading to cell death. Ag-NPs have a strong affinity for sulphur, phosphorous, and nitrogen molecules, affecting protein binding, DNA replication, and bacterial viability [25].
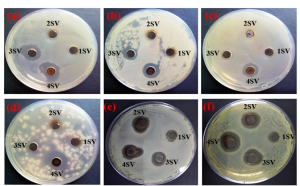
Fig 6: (a-f) displays the antimicrobial potential of Ag-NPs at varying concentrations of extract (10-40 vol%) at 0.75 μg.mL-1 against various bacterial pathogens, including Bacillus anthracis, Bacillus amyloliquefaciens, Staphylococcus aureus, S. warneri, E. coli, and Acinetobacter baumannii
3.4 Utilization of Ag-NPs from A. indica for the Lessening of MB and CR:
The time-dependent UV–vis spectra for the reduction of dyes are shown in Figure 7 (a–f). These spectra include dye with NaBH4 (a,d), dye with NaBH4 + A. indica extract (b,e), and dye with NaBH4 + Ag-NPs from A. indica (c,f). In addition, spectrophotometric analysis was performed to evaluate the catalytic activity of Ag-NPs derived from A. indica for the reduction of MB and CR in the presence of NaBH4. The Ag-NPs reduced MB and CR efficiently. Their catalytic performance is comparable to other plant-mediated Ag-NPs reported in literature, which typically show similar rapid electron-transfer behavior in the presence of NaBH After forty minutes, the reducing capacity of NaBH4 with dye is shown in Figure 7 (a,d). The addition of extract (A. indica) with NaBH4 does not have a significant impact on the reduction (b,e). The catalytic effectiveness of Ag-NPs derived from A. indica in conjunction with NaBH4 demonstrates a rapid and consecutive reduction in the concentration of dyes (c,f). Figure 7c illustrates the effective reduction of MB to leucomethylene blue in the presence of NaBH4 and nanocatalyst (Ag-NPs developed from A. indica) at room temperature. The decolorization process was recorded over a span of thirteen minutes, accompanied by a progressive decrease in absorbance spectra at a wavelength of λmax 663 nm [25]. The colorant CR dye had a pronounced peak at around 493 nm, accompanied by a shoulder at 350 nm. Despite this, the peak intensity steadily dropped despite the addition of NaBH4 and nanocatalyst over the course of one to nine minutes at room temperature. These Ag-NPs derived from A. indica have the potential to be exploited in the industry as nanocatalysts since they performed very well in the reduction of dyes via catalytic processes.
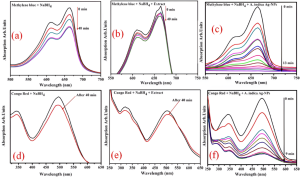
Fig 7: Time-dependent UV–vis spectra for the reduction of dyes (a–f). These spectra include dye with NaBH4 (a,d), dye with NaBH4 + A. indica seed extract (b,e), and dye with NaB H4 + Ag-NPs from A. indica (c,f).
Conclusion
This study successfully synthesized stable silver nanoparticles (Ag-NPs) using a green method with Setaria verticillata (SV) and Azadirachta indica (AI) seed exudates as reducing and capping agents. Comprehensive characterization using UV-Vis spectrophotometry, and FTIR confirmed their physicochemical properties. The Ag-NPs demonstrated significant antimicrobial activity, with AI-derived nanoparticles exhibiting superior efficacy against tested pathogens. Toxicity testing on earthworms indicated substantial inhibition, highlighting potential biological applications. Additionally, the Ag-NPs acted as effective nano-catalysts for the degradation of methylene blue (MB) and Congo red (CR) using NaBH₄, demonstrating their potential for eco-friendly dye removal. Possible applications such as anticancer and anthelmintic systems are proposed as future research directions rather than outcomes of the present work.
Author’s Contribution: S. B., Conceived the idea; L. A. Z, Designed the simulated work and the acquisition of data; S.B., simulated work, data analysis or analysis and interpretation of data and wrote the basic draft; R.A.A, Did the language and grammatical edits. A. J. L. provided critical feedback and helped shape the research; S. B. wrote the final version of the manuscript.
Funding: The publication of this article was funded by no one.
Conflicts of Interest: The authors declare no conflict of interest.
Acknowledgement: The authors would like to thank Chemistry department SBBU for their laboratory support during this research.
REFERENCES
[1] M.-C. Daniel and D. Astruc, “Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology,” Chem. Rev., vol. 104, no. 1, pp. 293–346, 2004.
[2] X. Ma, J. Geiser-Lee, Y. Deng, and A. Kolmakov, “Interactions between engineered nanoparticles (ENPs) and plants: phytotoxicity, uptake and accumulation,” Sci. Total Environ., vol. 408, no. 16, pp. 3053–3061, 2010.
[3] M. Singh, S. Manikandan, and A. K. Kumaraguru, “Nanoparticles: a new technology with wide applications,” Res. J. Nanosci. Nanotechnol., vol. 1, no. 1, pp. 1–11, 2011.
[4] W. Zhang and G. Wang, “Research and development for antibacterial materials of silver nanoparticle,” New Chem Mater, vol. 31, no. 2, pp. 42–44, 2003.
[5] L. S. Nair and C. T. Laurencin, “Silver nanoparticles: synthesis and therapeutic applications,” J. Biomed. Nanotechnol., vol. 3, no. 4, pp. 301–316, 2007.
[6] R. Nair, S. H. Varghese, B. G. Nair, T. Maekawa, Y. Yoshida, and D. S. Kumar, “Nanoparticulate material delivery to plants,” Plant Sci., vol. 179, no. 3, pp. 154–163, 2010.
[7] S. Iravani, H. Korbekandi, S. V. Mirmohammadi, and B. Zolfaghari, “Synthesis of silver nanoparticles: chemical, physical and biological methods,” Res. Pharm. Sci., vol. 9, no. 6, p. 385, 2014.
[8] G. Merga, R. Wilson, G. Lynn, B. H. Milosavljevic, and D. Meisel, “Redox catalysis on ‘naked’ silver nanoparticles,” J. Phys. Chem. C, vol. 111, no. 33, pp. 12220–12226, 2007.
[9] C. Haefeli, C. Franklin, and K. Hardy, “Plasmid-determined silver resistance in Pseudomonas stutzeri isolated from a silver mine,” J. Bacteriol., vol. 158, no. 1, pp. 389–392, 1984.
[10] P. Mukherjee et al., “Fungus-mediated synthesis of silver nanoparticles and their immobilization in the mycelial matrix: a novel biological approach to nanoparticle synthesis,” Nano Lett., vol. 1, no. 10, pp. 515–519, 2001.
[11] A. K. Jha, K. Prasad, K. Prasad, and A. R. Kulkarni, “Plant system: nature’s nanofactory,” Colloids Surfaces B Biointerfaces, vol. 73, no. 2, pp. 219–223, 2009.
[12] I. Kokina, V. Gerbreders, E. Sledevskis, and A. Bulanovs, “Penetration of nanoparticles in flax (Linum usitatissimum L.) calli and regenerants,” J. Biotechnol., vol. 165, no. 2, pp. 127–132, 2013.
[13] U. B. Jagtap and V. A. Bapat, “Green synthesis of silver nanoparticles using Artocarpus heterophyllus Lam. seed extract and its antibacterial activity,” Ind. Crops Prod., vol. 46, pp. 132–137, 2013.
[14] E. D. R. Angelina, R. Bavyaa, and R. Rajagopal, “Green synthesis and characterization of silver nanoparticles using Fenugreek seed extract,” Int J Sci Res Pub, vol. 3, no. 7, pp. 1–3, 2013.
[15] T. Čoja, K. Zehetner, A. Bruckner, A. Watzinger, and E. Meyer, “Efficacy and side effects of five sampling methods for soil earthworms (Annelida, Lumbricidae),” Ecotoxicol. Environ. Saf., vol. 71, no. 2, pp. 552–565, 2008.
[16] V. K. Vidhu and D. Philip, “Catalytic degradation of organic dyes using biosynthesized silver nanoparticles,” Micron, vol. 56, pp. 54–62, 2014.
[17] A. K. Singh, M. Talat, D. P. Singh, and O. N. Srivastava, “Biosynthesis of gold and silver nanoparticles by natural precursor clove and their functionalization with amine group,” J. Nanoparticle Res., vol. 12, pp. 1667–1675, 2010.
[18] Y. Liu, P. Laks, and P. Heiden, “Controlled release of biocides in solid wood. III. Preparation and characterization of surfactant‐free nanoparticles,” J. Appl. Polym. Sci., vol. 86, no. 3, pp. 615–621, 2002.
[19] H. Mansur, R. Oréfice, M. Pereira, Z. Lobato, W. Vasconcelos, and L. Machado, “FTIR and UV‒vis study of chemically engineered biomaterial surfaces for protein immobilization,” Spectroscopy, vol. 16, no. 3–4, pp. 351–360, 2002.
[20] S. M. Hussein, O. H. Shihab, and S. S. Ibrahim, “Interaction between Kaolin and DMSO: FTIR, XRD, thermodynamic and Nano studies,” J. Univ. Anbar Pure Sci., vol. 8, no. 1, 2014.
[21] N. A. Luechinger, R. N. Grass, E. K. Athanassiou, and W. J. Stark, “Bottom-up fabrication of metal/metal nanocomposites from nanoparticles of immiscible metals,” Chem. Mater., vol. 22, no. 1, pp. 155–160, 2010.
[22] M. R. Khan, M. A. Urmi, C. Kamaraj, G. Malafaia, C. Ragavendran, and M. M. Rahman, “Green Synthesis of Silver Nanoparticles with Its Bioactivity, Toxicity and Environmental Applications: A Comprehensive Literature Review,” Environ. Nanotechnology, Monit. Manag., p. 100872, 2023.
[23] D. J. Kvitek, J. L. Will, and A. P. Gasch, “Variations in stress sensitivity and genomic expression in diverse S. cerevisiae isolates,” PLoS Genet., vol. 4, no. 10, p. e1000223, 2008.
[24] D. R. Monteiro, L. F. Gorup, A. S. Takamiya, A. C. Ruvollo-Filho, E. R. de Camargo, and D. B. Barbosa, “The growing importance of materials that prevent microbial adhesion: antimicrobial effect of medical devices containing silver,” Int. J. Antimicrob. Agents, vol. 34, no. 2, pp. 103–110, 2009.
[25] P. Dibrov, J. Dzioba, K. K. Gosink, and C. C. Häse, “Chemiosmotic mechanism of antimicrobial activity of Ag+ in Vibrio cholerae,” Antimicrob. Agents Chemother., vol. 46, no. 8, pp. 2668–2670, 2002.