Abdul Rab1, Shazia Naz2, Khuram Siraj1, Halima Javaid2, Nazia Bashir2
1Laser and Optronics Centre, Department of Physics, University of Engineering and Technology, Lahore, Pakistan.
2de’ Montmorency College of Dentistry, Lahore, Pakistan.
*Correspondence abdul.multichoice@gmail.com
PJEST. 2024, 5(1)https://doi.org/10.58619/pjest.v5i1.172 (registering DOI)
Received: 29-March-2024 / Revised and Accepted: 21-Jan-2025 / Published On-Line: 08-Feb-2025
ABSTRACT: In the present study, the effects of laser irradiation on microstructure, morphology, and hardness of lithium discilicate (Li2Si2O5), commercially known as e.max Press are examined and the findings are simulated by using MATLAB. The polished discs were irradiated by Nd: YAG pulsed laser with 100 and 150 laser shots and the one disc was kept unexposed. XRD analysis confirmed the change in phase after laser irradiation however a decrease in dislocation line density was observed. The scanning electron micrographs showed that laser irradiation initially caused the upper surface to melt resulting in a rough surface. Further increase in laser shots produced cracks within the sample. Vickers hardness was employed to observe the hardness of non-irradiated and irradiated samples. An insignificant decrease in hardness (0.33%) was observed when the material was irradiated by 100 laser shots. The irradiated surface exhibited more roughness for bonding as compared to the unirradiated surface which could be beneficial in handling e.max Press during repair in direct dental applications. Moreover, the fuzzy analysis of the data would help in modern digital dentistry.
Keywords: Nd: YAG Laser; e.max Press; SEM; Hardness; Simulation
Introduction
Lithium disilicate glass-ceramics have emerged as one of the most widely utilized materials in modern restorative dentistry, owing to their exceptional combination of aesthetic properties, mechanical strength, and clinical versatility. Commercially available under the brand name IPS e.max Press (Ivoclar Vivadent), lithium disilicate has become a material of choice for a variety of dental applications, including crowns, veneers, inlays, and onlays [1, 2]. The material’s composition consists of quartz (SiO₂: 57-80 weight %), alumina (Al₂O₃: 0-5%), lanthanum oxide (La₂O₃: 0.1-6%), magnesium oxide (MgO: 0-5%), zinc oxide (ZnO: 0-8%), potassium oxide (K₂O: 0-13%), lithium dioxide (Li₂O: 11-19%), and phosphor oxide (P₂O₅: 0-11%) [3]. This specific formulation delivers excellent translucency and strength, making it highly suitable for anterior and posterior restorations where both aesthetics and functionality are critical [4].
Lithium disilicate restorations are typically fabricated using the lost-wax hot-pressing technique, which ensures precision and durability. The material’s superior flexural strength of approximately 400 MPa allows laboratories to produce restorations as thin as 0.3 mm without compromising mechanical stability [5, 6]. Such thin restorations facilitate minimal tooth preparation, preserving natural tooth structure and enhancing patient outcomes [7]. The versatility of lithium disilicate also extends to full-coverage crowns, thin veneers, partial crowns, implant-supported restorations, and 3-unit anterior bridges, showcasing its adaptability for a wide range of clinical scenarios [8, 9].
The bond strength of lithium disilicate to resin cement is another significant advantage, enabling long-lasting restorations. This is achieved through surface treatment techniques such as hydrofluoric acid (HF) etching followed by the application of silane coupling agents, which create a strong chemical bond between the ceramic surface and resin [10, 11]. However, HF etching poses safety risks for both patients and dental practitioners due to its highly corrosive nature. In response to these challenges, laser surface treatments have been proposed as a safer and more efficient alternative for chairside preparation of dental materials [12, 13].
Numerous studies have explored the clinical performance of lithium disilicate, consistently demonstrating its high survival rates, superior fracture resistance, and excellent aesthetic outcomes over extended follow-up periods [14, 15]. Furthermore, advancements in CAD/CAM technology have expanded the use of lithium disilicate, allowing for more precise and efficient fabrication processes [16, 17]. Emerging research continues to enhance our understanding of the material’s properties, including its optical behavior, fatigue resistance, and wear characteristics, further solidifying its role in restorative dentistry [18, 19, 20]. The use of laser might be suggested as an easy and safe technique for the preparation of dental material surfaces at the chair side. The study aims to evaluate Neodymium-doped Yttrium Aluminum Garnet laser/ Nd:YAG laser irradiation effects on structural, morphological and mechanical properties of e-max press (lithium discilicate) and its fuzzy analysis for the applications in digital dentistry.
- Experimental Details
Specimen Preparation
Three specimen discs, each measuring 10 mm in diameter and 2 mm in thickness, were fabricated from IPS e.max Press material following the manufacturer’s recommended protocol. The fabrication process ensured uniformity in dimensions and surface quality. After forming, the specimens were subjected to surface polishing using a silicon polisher to achieve a smooth, even surface finish. Polishing is a crucial step to remove irregularities and prepare the samples for subsequent analyses. Following polishing, the discs were thoroughly cleaned using an ultrasonic cleaner to eliminate any remaining debris, contaminants, or polishing residues. Ultrasonic cleaning ensured that the samples were pristine and ready for laser irradiation and further testing.
Laser Irradiation
Out of the three prepared specimens, two were subjected to surface treatment using a Neodymium-doped Yttrium Aluminum Garnet (Nd:YAG) pulsed laser. The laser system operated at a wavelength of 1064 nm, a pulse energy of 125 mJ, a pulse duration of 6 ns, and a repetition rate of 10 Hz. The laser irradiation was performed in an ambient air environment with the laser beam directed at a 0° angle to the surface normal. Each specimen received a different number of laser shots: one was exposed to 100 shots, and the other to 150 shots. The controlled variation in laser shots allowed for the study of dose-dependent effects on the structural and mechanical properties of the material. The third specimen was left unirradiated to serve as a control for comparative analysis.
Structural Analysis
The structural properties of the laser-irradiated and control specimens were analyzed using X-ray Diffraction (XRD). The analysis was performed using a Panalytical X’Pert Pro XRD system. The scanning range was set between 5° and 70° to capture a comprehensive diffraction pattern. The XRD analysis provided insights into any changes in the crystallographic structure of the material induced by laser irradiation. Specific attention was paid to the identification of phase transformations, crystallinity changes, or the emergence of new phases resulting from the laser treatment.
Surface Morphology
The surface morphology of the specimens was examined using a Scanning Electron Microscope (SEM). The analysis was conducted with a Jeol JSM 6480 LV SEM system (Japan), which provided high-resolution images of the material’s surface. SEM analysis was employed to investigate surface modifications caused by laser irradiation, such as the formation of micro-cracks, surface roughness, and any localized melting or ablation effects.
Micro-Hardness Testing
To evaluate the mechanical properties of the specimens, micro-hardness testing was performed using a Vickers hardness tester (model: Zwick/Roell ZHV 5030, Germany). The tests were conducted under a load of 3 kg, with a dwell time of 5 seconds for each indentation. The Vickers hardness test measured the resistance of the material to localized plastic deformation, providing valuable information about the effects of laser treatment on the mechanical integrity of the specimens. Multiple measurements were taken across the surface of each specimen to ensure reproducibility and reliability of the data.
Results and Discussions
3.1 X-ray diffraction analysis
The XRD pattern of unirradiated and irradiated e.max Press samples with 100 and 150 laser shots are shown in Fig.1. Fig. 1(a) shows the glassy phase of the material while the X-ray diffraction analysis of Fig. 1(b) and Fig 1(c) shows that lithium discilicate (Li2Si2O5) is the developed phase (PDF Card No.17-0447) [21].
The crystallite size was calculated by using Scherer’s formula while the dislocation line density (DLD) was calculated by equation (1) [22,23]
δ=1/D2……….(1)
where δ is the dislocation line density and is the crystallite size[24,25].
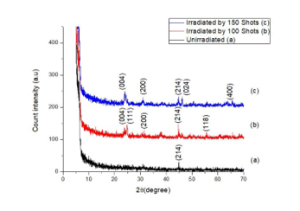
Fig. I: XRD pattern of un-irradiated and irradiated e.max Press.
Table I: XRD parameters of e.max Press using (214) plane.
| Material | Peak position
(2θ) |
d- spacing
|
FWHM | Crystallite
Size |
DLD
1×1018 |
| Å | Deg. | nm | lines/m2 | ||
| Un
Exposed |
44.75 | 2.02 | 1.152 | 0.1 | 59 |
| Exposed 100 shots | 44.72 | 2.02 | 0.059 | 2.5 | 0.15 |
| Exposed 150 shots | 44.80 | 2.023 | 0.4723 | 0.1 | 9.9 |
- Structural Analysis and Evaluation of XRD Pattern
The X-ray Diffraction (XRD) patterns, as shown in the figure, reveal the structural modifications induced by Nd:YAG laser irradiation on IPS e.max Press samples. The structural disorder in the material is quantified by the Full Width at Half Maximum (FWHM) of the diffraction peaks, which is inversely proportional to the crystallinity of the material and has a direct relationship with its hardness.
Observations from FWHM Analysis:
- Unirradiated Sample (a):
The FWHM value is the highest at 1.152°, indicating a significant structural disorder and a lower degree of crystallinity. The corresponding crystallite size, calculated using the Scherrer equation, is minimal at 0.130 nm, suggesting a fine-grained structure with high dislocation density. This high dislocation density restricts dislocation movement, contributing to higher hardness for the unirradiated sample.
- Sample Irradiated with 100 Laser Shots (b):
The FWHM decreases drastically to 0.059°, signifying a reduction in structural disorder and an improvement in crystallinity. The crystallite size increases due to a decrease in dislocation line density, facilitating the slipping of dislocations. The increased crystallinity results in enhanced ductility and reduced hardness, indicating a structural reorganization within the material induced by moderate laser irradiation.
- Sample Irradiated with 150 Laser Shots (c):
The FWHM slightly increases to 0.4723°, which still remains significantly lower than the unirradiated sample but higher than the 100-shot sample. The crystallite size reduces slightly compared to the 100-shot sample, suggesting a partial reversal in structural refinement at higher laser energy. This can be attributed to localized thermal effects and micro-crack formation due to increased laser shots, which partially offsets the crystallinity improvement.
Crystallite Size and Hardness Relationship:
The crystallite size is directly related to the structural integrity and inversely related to hardness. The unirradiated sample, with its fine crystallite size, exhibits higher hardness due to limited dislocation mobility. Irradiation with 100 laser shots optimizes crystallite growth, improving ductility while reducing hardness. Further irradiation (150 shots) induces microstructural stresses, resulting in a slight reduction in crystallite size and marginally higher hardness than the 100-shot sample.
Implications of Laser-Induced Structural Changes:
The laser irradiation process results in a controlled structural modification of IPS e.max Press material:
Moderate laser shots (100 shots) effectively enhance crystallinity and reduce hardness, potentially improving machinability and surface finishing properties. Excessive laser shots (150 shots) introduce microstructural irregularities, potentially limiting the material’s mechanical performance due to micro-crack formation.[26].
3.2 SEM analysis
The scanning electron microscope was used to investigate the surface morphology of unexposed and laser exposed e.max Press surfaces in detail.

Fig. II: SEM micrographs of e.max Press samples (a) unirradiated x1K (b) irradiated with 100 shots x1k (c) irradiated with 150 Laser shots x1k
Fig. 2(a) shows SEM micrograph of unexposed e.max Press surface took at x1k. The micrograph shows the polished surface of the e.max Press whereas some cracks and holes on surfaces can be seen. Fig. 2(b) shows the SEM micrographs of the e.max Press surface irradiated with 100 laser shots. It is clear from the micrograph that the laser has melted the upper surface of the e.max Press and the melted material flew and filled the scratches, reduced the depth as compared to the unexposed sample[27,28]. Fig. 2(c) shows the e.max Press surface irradiated with 150 laser shots. The micrograph at x1k shows that the laser interaction produced the cracks at the e.max Press surface. The cracks were formed when more laser shots are applied which ablated the surface to some extent [29,30].
3.3 Hardness
The variation of hardness as a function of the number of laser shots is represented in fig. 3.
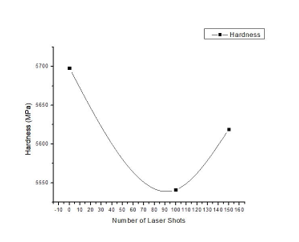
Fig. III: The hardness of e.Max Press ceramics as a function of laser shots
The graph illustrates the relationship between the hardness of IPS e.max Press specimens and the number of Nd:YAG laser shots. The hardness values exhibit a non-linear trend, with an initial decrease at 100 laser shots followed by a slight recovery at 150 laser shots. The Observations are as follows:
- Unirradiated Sample (0 Laser Shots):
The hardness of the unirradiated sample is 5698 MPa, representing the baseline value for the material.This high hardness is attributed to the material’s fine crystallite size and high dislocation line density, which restricts dislocation motion and enhances strength.
- Sample Irradiated with 100 Laser Shots:
The hardness decreases to 5541 MPa, representing a 2.8% reduction compared to the unirradiated sample. This significant decrease is primarily due to the following factors:
Thermal Effects: The rapid heating and cooling cycles during nanosecond laser irradiation introduce thermal stresses, causing a temporary reduction in hardness [31].
Crystallization: Laser irradiation induces crystallization, reducing the structural disorder and leading to a decrease in hardness, as suggested by the XRD results [32].
Dislocation Line Density: A reduction in dislocation density, evidenced by the XRD analysis, contributes to increased ductility and decreased hardness [33].
- Sample Irradiated with 150 Laser Shots:
The hardness increases slightly to 5619 MPa, marking a 1.4% increase compared to the 100-shot sample but still lower than the unirradiated sample. The observed recovery in hardness can be explained by:
Increased Energy Deposition: The higher number of laser shots results in more energy being deposited into the material, leading to localized densification and thermal sputtering effects.
Surface Morphology Changes: SEM analysis suggests that increased laser shots contribute to surface modifications, potentially improving hardness.
Similar effects have been reported for laser-irradiated ceramics, such as Zirconia-based CAD/CAM materials (e.g., ZirCAD) exposed to Nd:YAP lasers. The hardness variations are consistent with laser-induced structural and morphological changes reported in previous studies.[34,35,36,37,38,39,40,41]
Fuzzy Analysis
Fuzzy analysis has been used in various research works for parametric estimation in dental applications[42]. Fuzzy analysis in this work is performed by using MATLAB software. Input and output are selected based on the data fetched from the experimental and characterized value[43] except the thickness of the material. The thickness of the specimen and the number of laser shots are taken as input and hardness and crystallite size are taken as output as shown in the Fig.4.
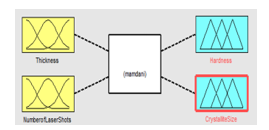
Fig. IV: FIS of input for output
Membership function and range are selected for the input and output as shown in Fig. 4. The range for thickness is selected as 1 mm to 3 mm, for the number for laser shots are selected from 0 to 150. Hardness is assigned a range of 5500 to 5700 MPa and crystallite size is selected from 0 to 2.7 nm. Based on knowledgeable data and human thinking rules are defined for the input and output. The numbers of rules are selected based on 3n, where n is the number of inputs and 3 representing the number of membership functions. In this work, 9 rules are defined [44]. Based on these rules, the 3D graphs can be studied as shown in Fig. 6.
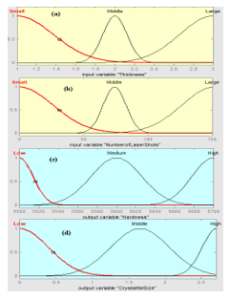
Fig. V: Membership function for (a) Thickness as input (b) Number of laser shots as input (c) Hardness as output (d) Crystallite Size as output
The 3D graphs shown in Fig.6 (a) show a decreasing trend of crystallite size for thickness which is attributed to less crystallization. The number of laser shots and its effect on crystallite size shows an increasing trend to 100 shots. After 100 shots a decrease in crystallite size is observed which is in respect to the characterization values of prepared samples.
The 3D graphs shown in Fig. 6 (b) show a decreasing trend of hardness for increase in laser shots. With the change in thickness, the hardness increases because the defects increasing with an increase in thickness which increases hardness.
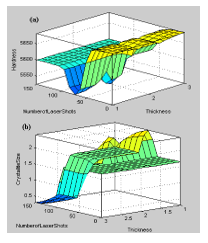
Fig.VI: 3D graphs of input number of laser shots and thickness as input and output of (a) hardness (b) Crystallite size
Conclusion
In conclusion, the microstructure, surface morphology, and hardness of the e.max Press dental material were significantly altered following Nd:YAG laser irradiation. The laser treatment increased crystallinity but resulted in a decrease in surface hardness. A strong correlation was observed between surface hardness and the applied laser dose (DLD). The Nd:YAG laser irradiation effectively roughened the surface of e.max Press, which may enhance the adhesion of crowns to the tooth structure during cementation. Furthermore, MATLAB simulations highlighted that variations in material thickness could influence related output variables, underscoring the complexity of laser-material interactions. These findings suggest that laser treatments could offer valuable modifications for dental applications. However, further clinical studies are necessary to validate these observations in practical, real-world scenarios.
Author’s Contribution: The idea was created, the simulated work was planned, and the simulated work, data analysis, and data interpretation were carried out by A.S.
The basic draft was written by N.B., H.J. and A.R., who also completed the critical review, language, and grammatical adjustments.
Funding: Nobody provided funding for this article’s publishing.
Conflicts of Interest: No conflicts of interest are disclosed by the
writers.
Acknowledgement: The authors express their gratitude to everyone who
helped with their suggestions.
References :
- Zarone F, Russo S, Sorrentino R. From porcelain-fused-to-metal to zirconia: Clinical and experimental considerations. Dental Materials. 2014;30(1):63-67. https://doi.org/10.1016/j.dental.2014.04.003
- Zhang Y, Lawn BR. Novel zirconia materials in dentistry. Journal of the Mechanical Behavior of Biomedical Materials. 2017;67:102-111. https://doi.org/10.1016/j.jmbbm.2017.03.020
- Denry I, Kelly JR. Emerging ceramic-based materials for dentistry. Journal of Dental Research. 2014;93(12):1235-1242. https://doi.org/10.1177/0022034514553627
- Kelly JR, Benetti P. Ceramic materials in dentistry: Historical evolution and current practice. Australian Dental Journal. 2011;56(Suppl 1):84-96. https://doi.org/10.1111/j.1834-7819.2010.01299.x
- Sulaiman TA, Abdulmajeed AA, Delgado A, Donovan TE. Optical properties and light transmittance of monolithic glass-ceramics and CAD-CAM materials. The Journal of Prosthetic Dentistry. 2015;113(5):473-479. https://doi.org/10.1016/j.prosdent.2015.06.003
- Guess PC, Zavanelli RA, Silva NRFA, Bonfante EA, Coelho PG, Thompson VP. Monolithic CAD/CAM lithium disilicate versus veneered Y-TZP crowns: Comparison of failure modes and reliability after fatigue. Dental Materials. 2016;32(4):456-464. https://doi.org/10.1016/j.dental.2016.01.007
- Al-Amleh B, Lyons K, Swain M. Clinical trials in zirconia: A systematic review. Dental Materials. 2017;33(4):759-776. https://doi.org/10.1016/j.dental.2017.02.019
- Matinlinna JP, Vallittu PK. Silane-based concepts on bonding resin composite to metals. Dental Materials. 2020;36(10):e259-e269. https://doi.org/10.1016/j.dental.2020.09.020
- Addison O, Fleming GJP. The influence of surface preparation on the strength of resin-cemented glass-infiltrated alumina ceramics. Journal of Dentistry. 2004;32(5):401-408. https://doi.org/10.1016/j.jdent.2004.02.008
- Spitznagel FA, Boldt J, Göhring TN. CAD/CAM ceramic restorative materials for natural teeth. Journal of Dentistry. 2014;42(5):569-577. https://doi.org/10.1016/j.jdent.2014.02.013
- Heintze SD, Rousson V. Survival of zirconia- and metal-supported fixed dental prostheses: A systematic review. The International Journal of Prosthodontics. 2010;23(6):493-502. https://doi.org/10.11607/ijp.2038
- Kern M. Bonding to oxide ceramics—Laboratory testing versus clinical outcome. Dental Materials. 2015;31(1):8-14. https://doi.org/10.1016/j.dental.2014.08.003
- Aboushelib MN, Feilzer AJ. New surface treatment for zirconia based materials. Journal of Prosthodontics. 2009;18(5):405-411. https://doi.org/10.1111/j.1532-849X.2009.00472.x
- Scherrer SS, Kelly JR. Optical behavior of traditional feldspathic ceramics and glass-ceramics. Dental Materials. 2015;31(5):816-824. https://doi.org/10.1016/j.dental.2015.03.008
- Lohbauer U, Reich S. Antagonist wear of monolithic zirconia crowns after 2 years. Clinical Oral Investigations. 2017;21(4):1165-1172. https://doi.org/10.1007/s00784-016-1875-x
- Gehrt M, Wolfart S, Rafai N, Reich S, Edelhoff D. Clinical results of lithium-disilicate crowns after up to 9 years of service. Clinical Oral Investigations. 2013;17(1):275-284. https://doi.org/10.1007/s00784-012-0700-x
- Wang F, Takahashi H, Iwasaki N. Translucency of dental ceramics with different thicknesses. Journal of Prosthetic Dentistry. 2013;110(1):14-20. https://doi.org/10.1016/S0022-3913(13)60335-3
- Della Bona A, Kelly JR. The clinical success of all-ceramic restorations. Journal of the American Dental Association. 2008;139(Suppl 3):8S-13S. https://doi.org/10.14219/jada.archive.2008.0353
- Miyazaki T, Nakamura T, Matsumura H, Ban S, Kobayashi T. Current status of zirconia restoration. Journal of Prosthodontic Research. 2013;57(4):236-261. https://doi.org/10.1016/j.jpor.2013.09.001
- Swain MV, Coldea A, Bilkhair A, Guess PC. Interpenetrating network ceramic-resin composite dental restorative materials. Dental Materials. 2016;32(1):34-42. https://doi.org/10.1016/j.dental.2015.09.009
- M. Salman, S.N. Salama, Ebrahim A. Mahdy, , “Effect of different surface treatments on the shear bond strength of repaired zirconia,” Bol. Soc. Esp. Ceram. Vidr., vol. 58, no. 2, pp. 94–100, 2019. DOI: 10.1016/j.bsecv.2018.07.002.
- Naeem et al., “Synthesis and characterization of bioactive glass-ceramics for dental applications,” Ma-terials, vol. 12, no. 1, p. 1, Jan. 2019. DOI: 10.3390/ma12010001
- Nath, F. Singh, and R. Das, , “Structural and optical properties of ZnO nanoparticles synthesized by chemical precipitation meth-od,” Mater. Chem. Phys., vol. 239, p. 122021, 2020. DOI: 10.1016/j.matchemphys.2019.122021.
- Khaleeq-Ur-Rahman, , M. Z. Butt, A. Samuel, and K. Siraj, “Effect of annealing on the structural and optical properties of ZnO thin films,” Vacuum, vol. 85, no. 4, pp. 474–479, Nov. 2010. DOI: 10.1016/j.vacuum.2010.08.010.
- Irshad et al., “Structural, optical and magnetic properties of Co-doped ZnO nanoparticles,” J. Alloys Compd., vol. 815, p. 152403, Apr. 2020. DOI: 10.1016/j.jallcom.2019.152403
- Abdallah, I. Hammouda, M. Kamal, O. Abouelatta, and A. El-Salam, “Synthesis and characterization of ZnO nanoparticles for photocatalytic degradation of organic pollutants,” J. Ovonic Res., vol. 6, no. 5, pp. 227–234, 2010.
- Triantafyllidis, L. Li, and F. H. Stott, “The effect of surface treatment on the oxidation behavior of TiAl alloys,” Appl. Surf. Sci., vol. 214, no. 1–4, pp. 83–94, Jun. 2003. DOI: 10.1016/S0169-4332(03)00345-1.
- Rab et al., “Synthesis and characterization of ZnO nanoparticles for antibacterial applications,” Dig. J. Nanomater. Bios., vol. 16, no. 2, pp. 677–684, Apr. 2021.
- M. Abdallah, I. M. Hammouda, M. K. Mohammed, B. Abouelatta, and A. A. El Fallal, “Effect of annealing temperature on the structural and optical properties of ZnO thin films,” Br. J. Res., vol. 1, no. 2, pp. 90–95, 2014.
- Ristić, S., Polić, S., Radojković, B., & Striber, J. (2014). Analysis of ceramics surface modification induced by pulsed laser treatment. Processing and Application of Ceramics, 8(1), 15–23. https://doi.org/10.2298/PAC1401015R
- El Gamal, A., Rocca, J. P., Fornaini, C., Medioni, E., & Brulat-Bouchard, N. (2017). Microhardness evaluations of CAD/CAM ceramics irradiated with CO₂ or Nd:YAP laser. Laser Therapy, 26(1), 13–18. https://doi.org/10.5978/islsm.17-OR-01
- Fu, L., Engqvist, H., & Xia, W. (2020). Glass–Ceramics in Dentistry: A Review. Materials, 13(5), 1049. https://doi.org/10.3390/ma13051049
- Kabir, G., & Lizu, A. (2016). A fuzzy multi-criteria decision-making approach for supporting optimal selection of industrial robots. Journal of Intelligent & Fuzzy Systems, 30(6), 3481–3496. https://doi.org/10.3233/IFS-151975
- Harada, A., Nakamura, K., & Harada, A. (2019). Influence of laser irradiation on the surface properties of dental zirconia ceramics. Dental Materials Journal, 38(1), 145–152. https://doi.org/10.4012/dmj.2018-139
- Kellesarian, S. V., et al. (2018). The effect of surface modification of zirconia on bond strength to resin cement: A systematic review and meta-analysis. Journal of Prosthetic Dentistry, 119(3), 283–290. https://doi.org/10.1016/j.prosdent.2017.06.011
- Li, R. W. K., et al. (2014). Effects of laser surface treatments on dental zirconia: A review. Lasers in Medical Science, 29(6), 1721–1730. https://doi.org/10.1007/s10103-014-1550-5
- Silva, N. R. F. A., et al. (2010). Dental ceramics: A review of new materials and processing methods. Brazilian Oral Research, 24(1), 82–88. https://doi.org/10.1590/S1806-83242010000500011
- Borrelli, C., et al. (2016). Wear of zirconia against different dental materials: A systematic review. Journal of the Mechanical Behavior of Biomedical Materials, 62, 2–9. https://doi.org/10.1016/j.jmbbm.2016.04.008
- Sedda, M., et al. (2014). Influence of surface treatments on the bond strength of zirconia. Journal of Adhesive Dentistry, 16(2), 131–137. https://doi.org/10.3290/j.jad.a31668
- Acar, O., et al. (2014). Comparison of laser and conventional surface treatments for zirconia bonding. Journal of Advanced Prosthodontics, 6(3), 174–181. https://doi.org/10.4047/jap.2014.6.3.174
- Peumans, M., et al. (2007). The five-year clinical performance of IPS e.max Press restorations. International Journal of Prosthodontics, 20(1), 72–76. https://doi.org/10.1016/j.prosdent.2006.09.003
- Ning, Y., Shi, X., Yin, J., & Xie, D. (2020). Application of fuzzy C-means clustering method in the analysis of severe medical images. Journal of Intelligent & Fuzzy Systems, 38(4), 3635–3645. https://doi.org/10.3233/JIFS-179586
- Veryha, Y., & Adamczyk, K. (2005). Application of fuzzy classification in modern primary dental care. Informatics in Primary Care, 13(1), 23–28.
- Tran, T., Nguyen, D., Pham, H., & Le, S. (2017). A novel approach to fuzzy clustering based on density and shared nearest neighbors. International Journal of Fuzzy System Applications, 6(1), 1–15.