Rabia Nawaz1*, Zaiba Naz1, Muhammad Saad Raza1, Zohal Hassan1, Attiya Razzaq1, Samia Afzal2, Urooj Irshad1, Ruqaya Khurshid1, Robina Yaseen1, Ghazala Rafique1, Uqba Mehmood1
1Department Biological Sciences, Superior University, Lahore, Pakistan
2Center of Excellence in Molecular Biology, University of the Punjab, Lahore, Pakistan
*Correspondence rabia.nawaz8@gmail.com
PJEST. 2024, 5(1) https://doi.org/10.58619/pjest.v5i1.174 (registering DOI)
Received: 28-Nov-2024 / Revised and Accepted: 20-Jan-2025 / Published On-Line: 08-Feb-2025
ABSTRACT: Protein leakage through urine, at a level exceeding 3.5g per 1.7m^2 of body area per day, is a symptom characteristic of Nephrotic syndrome. It’s a diverse condition with symptoms including edema, hypoalbuminemia (≤ 25g/L), albumin excretion greater than 40 mg/m^2 per hour, uPCR ≥ 2,000 mg/g or 3+ protein in urine test. Springtime is when this syndrome is more common. A detailed literature review was conducted by using credible databases and scientific journals. This review concludes that in nephrotic syndrome, which is caused by metabolic and genetic defects, patients have to face proteinuria, edema, hyperlipidemia, and hypoalbuminemia. This study additionally focuses on the use of corticosteroids to treat nephrotic syndrome and the proper doses needed for effective therapy. Our study identified the complications related to nephrotic syndrome including the syndrome itself and post-treatment complications. Corticosteroids have been reported to side effect and relapse of nephrotic syndrome has also been identified as a health issue.
Keywords: Nephrotic syndrome, corticosteroids, steroid-resistant nephrotic syndrome prednisolone, levamisole, cyclophosphamide.
Introduction
Nephrotic syndrome is characterized by a condition in which protein is leaked through urine and its level rises above 3.5g per 1.7 of the body area per day [1]. It is a heterogeneous disorder characterized by edema, uPCR ≥ 2,000 mg/g or 3+ protein in urine test, hypoalbuminemia (≤ 25g/L), and albumin excretion greater than 40mg/ per hour [2]. This syndrome is more widespread in spring season [3].
The kidney’s glomerular filtration barrier, consisting of the glomerular membrane and podocyte-layered epithelium, prevents protein loss [4]. Disruptions due to genetic defects, metabolic disorders, or infections lead to proteinuria [5]. Alterations in podocytes are main contributors to this dysfunction [6,7,8,9,10].
Illustration of nephrotic syndrome is shown in Figure 1.
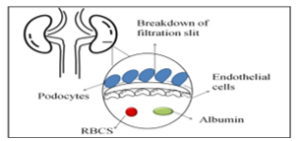
Fig. I: Representation of Nephrotic syndrome (The human kidney is consisted of various layers and glomerular filtration barrier; the outer layer is epithelial layer made up of podocytes which encompassed the inner layer of endothelial cells. The red blood cells (RBCS) and albumin proteins are present under the layer of endothelial cells. The breakdown of the filtration slit bridged between podocytes causes the leakage of protein and red blood cells in urine leading to nephrotic syndrome.)
Classification
Nephrotic syndrome can be primary or secondary, almost 10% cases are of secondary nephrotic syndrome and 90 % are of primary nephrotic syndrome. Nephrotic syndrome is primarily classified into three major classes. These types include Minimal change nephrotic syndrome (MCNS), Focal segmental glomerulosclerosis (FSGS) and Membranous nephropathy (MN) [11]. The detail of primary nephrotic syndrome and causes of secondary nephrotic syndrome is shown in Figure 2.
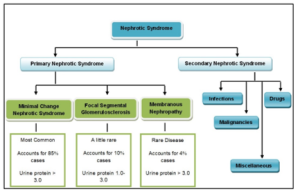
Fig. II: Classification of nephrotic syndrome and their descriptions (Nephrotic syndrome is categorized into primary nephrotic syndrome and secondary nephrotic syndrome. The primary nephrotic syndrome is further categorized into Minimal change nephrotic syndrome that accounts for 85% cases, Focal segmental glomerulosclerosis accounting for 10% cases and Membranous Nephropathy that account for 4% cases; the causes of Secondary Nephrotic syndrome include infections, malignancies, miscellaneous and drugs).
Causes
Exact nephrotic syndrome’s causes are still unknown but according to some studies it involves immune dysregulation and structural abnormalities of the podocytes that are inherited [12]. Early substantiation supported the disproportion of the T-helper responses [13] but the later studies show proofs of impaired T regulatory function, which also suggest variations in adaptive and innate immune responses [14]. In adults of European countries, nephrotic syndrome is caused by membranous nephropathy which affects the filter of kidney and reduces kidney’s function. In countries of Africa, nephrotic syndrome is reported to be caused because of focal segmental glomerulosclerosis [15].
Evaluation of Nephrotic Syndrome
Analysis of nephrotic syndrome is evaluated by family and medical history, physical exam, urine tests, blood tests, renal biopsy and ultrasonography. In urine tests, urine sample of 24 hours is examined in which proteinuria is diagnosed [15, 16]. The methods of evaluation of nephrotic syndrome along with albumin, triglyceride and cholesterol ranges are given in detail in Table I.
Table I: Methods for evaluation of nephrotic syndrome along with their standard ranges and indications.
| Evaluation of Nephrotic Syndrome | |||
|
Urine Tests
|
Proteinuria Range | Other Indications | |
|
3+ or 4+ reading on dipstick |
Urine test can indicate the presence of casts, fats, epithelial cell and lipids which shows glomerular disorder and also presents creatinine ratio. | ||
|
Blood Tests
|
Serum Albumin | Cholesterol & Triglyceride level | Other Indications |
| Less than 2.5g/dL | It is highly increased if NS is present. | Serological tests, Rheumatoid factor, cryoglobulins results indicate the secondary cause of nephrotic syndrome | |
| Ultrasonography | It demonstrates renal echogenicity | ||
| Kidney Biopsy | Examination of kidney by taking small piece of kidney tissue | ||
Complications of Nephrotic Syndrome
Acute and chronic renal failure is the major problem of nephrotic syndrome. Bilateral renal vein thrombosis can lead to acute renal failure whose diagnosis can be done by ultrasonography [17]. Brain, peripheral vessels and lungs can be affected by these complications which are caused due to dropping of protein in the urine. In nephrotic syndrome, immune system starts attacking its own cells [18]. It also includes the reduction in plasma IgG synthesis due to its loss in urine that can cause bacterial infections [19]. High temperature and abdominal pain are reported in 80% of the patients having nephrotic syndrome [20]. Cellulitis is a serious bacterial skin infection, is also reported in patients. It usually appears on the lower legs, in it the skin becomes swollen, red and painful [21]. Meningitis is also one of the problems, in which the protective membranes of brain and spinal cord become inflamed [22]. In nephrotic syndrome, immense retention of water and sodium is also a contributing factor towards complications [23]. Protein loss through the urine is the major complication. In this syndrome, the whole body becomes swollen due to inflammations in many parts. Edema, swelling in legs, feet, hands and face can be caused by sodium retention [24]. The urinary loss of albumin and loss of vitamin D binding protein, which causes vitamin D deficiency leading to bones weakness, has also been reported [25]. Cerebral hemorrhage due to biochemical abnormalities is a severe complication that has been reported in the patients of nephrotic syndrome [26]. Risks of heart attack in nephrotic syndrome patients because of hypertension are also elevated [27].
Nephrotic Syndrome in Children
Nephrotic syndrome mostly affects children, adults also suffer from it but very few cases have been cited so far. In children mostly MCNS and FSGS nephrotic syndrome occurs, MN is quite rare in children [28]. In children, idiopathic nephrotic syndrome (INS) is the most common glomerular disease, with corticosteroids as the first-line treatment. Most children respond well to steroids and are reported as having steroid-sensitive nephrotic syndrome, which typically causes frequent relapses, requiring repeated steroid cycles which can lead to significant side effects. In contrast, children who do not respond to steroids are diagnosed with steroid-resistant nephrotic syndrome, which may require second-line immunosuppressants, such as calcineurin inhibitors, to help control the disease. SR nephrotic syndrome can progress to multidrug resistance, often resulting in renal failure and a higher risk of relapse after a kidney transplant. For long-term side effects and to minimize complications, close monitoring of the patient’s condition and steroid-sparing agents, i.e. levamisole, mycophenolate mofetil, and anti-CD20 monoclonal antibodies are used, to avoid severe complications and ensure significant treatment [29]. In children affected from nephrotic syndrome, hyperactive platelets are observed. Most of the affected children have steroid – sensitive nephrotic syndrome and about 20% of children have steroid-resistant nephrotic syndrome [30]. Nephrotic syndrome which manifests at birth, known as congenital nephrotic syndrome, is acute and non-treatable [31].
Nephrotic Syndrome in Adults
Bacterial peritonitis with nephrotic syndrome is not common in adults [32], only 14 cases have yet been reported [3]. In teenage groups, there is no specific difference between genders but in younger groups, nephrotic syndrome is more common in boys than girls [33]. Edema is a common complication of nephrotic syndrome that requires diuretic treatment. However, factors such as poor drug compliance, renal perfusion issues, changes in renal function, and electrolyte imbalances can lead to diuretic resistance (DR). DR can result in higher mortality, increased readmission rates, and ineffective diuretic use. Managing hypervolemia is crucial, as proteinuria and hypoalbuminemia cause fluid extravasation, triggering neurohormonal mechanisms that increase salt and water retention. While diuretics and human-albumin infusion were traditionally recommended, recent studies show a kidney-limited sodium-reabsorption mechanism activated by serine proteases and ENaC channels. Loop diuretics remain the first-line treatment, but patients may develop resistance due to factors like kidney dysfunction, poor compliance, and high-sodium diets. Overcoming DR may involve adding new diuretics, ultrafiltration, adjusting diuretic doses, or using sequential nephron blockade. ENaC blockers have been reported as promising potential treatments for nephrotic edema. Clinicians should focus on addressing the underlying causes of DR to prevent complications and improve treatment outcomes in adult nephrotic syndrome patients [34].
Therapeutics for Nephrotic Syndrome
Corticosteroids and non-corticosteroids both can be used to treat nephrotic syndrome. Both treatment approaches have its beneficial and side-effects on the patients [35]. The treatment along with benefits and drawbacks of non-corticosteroids and corticosteroids are discussed in detail as follows:
Non-Corticosteroids:
At early phases, non-corticosteroid therapies for nephrotic syndrome have received limited attention. To date, only a few trials have looked into these alternatives. The only published trial that focused on a non-corticosteroid treatment during the initial phase was a noteworthy study by Hoyer and Brodehl (2006), which demonstrated that ciclosporin did not change the development of the disease when compared to conventional corticosteroid dose. Ongoing research, however, attempts to close this gap by assessing the possible early intervention benefits of medications often used in relapsing disease. Studies are being conducted to evaluate alternatives to corticosteroids. Initiated in 2016, the multi-center, placebo-controlled, double-blind LEARNS and NEPHROVIR3 (NCT02818738) trials examine the use of levamisole at the onset of idiopathic nephrotic syndrome. A low-risk immunomodulatory drug called levamisole exhibits promise for its ability to alter illness. The effectiveness of early mycophenolate mofetil (MMF) administration in comparison to conventional corticosteroid therapy is being assessed by the open-label RCT known as the INTENT trial. Furthermore, the RITURNS study supports the use of rituximab as the first steroid-sparing agent, with additional confirmation expected from a Japanese placebo-controlled trial. A French study is currently evaluating rituximab as a first-line treatment for adult minimal change nephropathy, which could inform pediatric approaches [36]. The non-corticosteroids treatment of nephrotic syndrome includes the diuretic drugs, ACE Inhibitors, statins, vaccines and adequate diet and supplements.
Diuretic Drugs
Diuretic drugs are firstly used for nephrotic syndrome, such as loop diuretics which prevent reabsorption of sodium in loop of Henle, so that loss of sodium can be reduced. Some factors disrupted in nephrotic syndrome, such as sodium intake, plasma protein levels, and renal perfusion, influence its effectiveness [35]. In 2015, Qavi and his coworkers recommended tailored diuretic strategies in nephrotic syndrome, including sequential nephron blockade with loop diuretics and distal-acting agents for refractory edema. They also recommend albumin infusions with loop diuretics in cases of severe hypoalbuminemia to improve renal diuretic delivery.
Diuretic resistance is a common complication of nephrotic syndrome, especially when there is severe hypoalbuminemia or impaired renal function. Resistance could result from compensatory mechanisms like increased sodium reabsorption in distal nephron segments or altered diuretic pharmacokinetics. To overcome resistance, consider using higher doses, combining diuretics (e.g., loop and thiazide diuretics), and addressing underlying causes such as high sodium intake or poor treatment adherence.
New therapeutic agents, such as ENaC blockers (e.g., amiloride), could be useful additions to diuretic regimens. These drugs target sodium reabsorption in the distal nephron and may be used as supplements for patients who do not respond to conventional treatments. Furthermore, ultrafiltration is being considered as an option for patients with severe, treatment-resistant hypervolemia. This therapy is effective mostly in children but only in those patients who are at very early stage. Heavy number of diuretic drugs can cause secondary renal failure and thromboembolism [37]. Diuretics basically reduce edema [38].
Angiotensin-Converting Enzyme Inhibitors (ACE Inhibitors)
Various studies have shown that ACE inhibitors are helpful in the reduction of proteinuria. ACE inhibitor and Angiotensin II receptor blockers are the important players of therapy against nephrotic syndrome [39]. Researchers suggest the treatment of 12 weeks with these two to reduce proteinuria. These two also reduce loss of podocytes and lower the risk of renal failure [11].
Adequate Diet and Supplements
Due to excessive loss of protein, sodium and water in patients of nephrotic syndrome, the nutritional imbalance prevails in most cases. To overcome this problem, adequate diet and proper intake of vitamins and additional nutrient supplements must be taken by the patients. Several studies suggest that, patients with nephrotic syndrome should be taking a diet which has low salt contents [40]. Diet as a therapy has been found effective to some extent but this lacks specificity and can have severe side effects because of nutritional imbalance in different cases [11].
Corticosteroids
Corticosteroids are the synthetic hormones. Corticosteroids are accessible in different versions like oral tablets, injections, gels and inhalers. They are used as anti-inflammatory drugs that prevent the production of inflammatory proteins. Corticosteroids reduce inflammation in the digestive tract; on the other hand, they lower the functioning of the immune system hence automatically reducing immunity against pathogens [41].
Management of Nephrotic Syndrome
Treating nephrotic syndrome with steroids was the crucial challenge in the 20th century [42]. Use of corticosteroids for the treatment of nephrotic syndrome depends on the choice of physician [43]. For treating nephrotic syndrome, detailed examination should be recorded like physical evaluation for detecting disorders and infections [44]. About 80% to 90%children respond to corticosteroids but 20-30% become steroid-dependent [45].
Corticosteroid therapy used for the treatment of children having steroid sensitive nephrotic syndrome has been observed to be more beneficial as it completely remits the syndrome but in case of steroid resistant nephrotic syndrome, children do not achieve complete remission from disease. In complete remission patient’s clinical tests show uPCR < 200mg/g or less then 1+ protein in urine test for 3 successive days [46], and partial remission is considered when uPCR is between 200 and 2000 mg/g and proteinuria reduces to 50% [47].
Treatment for the Early-Stage Steroid-Sensitive Nephrotic Syndrome
Kidney Disease Improving Global Outcomes (KDIGO) workgroup in 2012 recommend the use of corticosteroid therapy for at least 12 weeks. They also suggested that oral prednisone is administered as a single daily dose that have to be started as 2mg/kg/day to a maximum 60mg/day and this has to be given for 4 to 6 weeks, followed by alternate day medication and continued for 2 to 5 months with reducing the dose. Studies recommend that daily prednisone should be given to children for at least 4 weeks based on randomized controlled trials (RCT) so the 8 weeks that contain 4 weeks of daily prednisone and 4 weeks of alternate day may prove remarkably more productive in sustaining remission at 6 and 12 months [48]. Children who don’t respond to steroids usually have FSGS nephrotic syndrome [49].
Management of Relapsing Steroid Sensitive Nephrotic Syndrome
Most of the children suffering from nephrotic syndrome respond effectively in the early ages but the repeated courses of steroids due to relapses, cause high steroid toxicity. Patients, who often undergo relapses, develop steroid dependent nephrotic syndrome, so an alternative treatment is required for them [50, 51]. According to an estimate 70 percent relapses in patients follow the upper respiratory tract infection [52]. Chinese medical herb “TIAOJINING” and “Astragalus Decoction” play role in preventing these infections and proteinuria, edema and other all symptoms will be solved after one year [52, 53]. 80 to 90 percent children affected from steroid sensitive nephrotic syndrome have chance of relapses [54]. Experimental studies suggest that low dose of prednisone to the patient on alternate days, which should be about 0.48mg/kg and daily prednisone of about 0.25mg/kg, reduces the risk of relapses. Kidney Disease Improving Global Outcomes (KDIGO) workgroup proposed alkylating agents for periodically relapsing nephrotic syndrome [55].
Drugs for preventing frequent relapses
The most beneficial and effective drugs for preventing frequent relapses are prednisolone, levamisole, cyclosporine, cyclophosphamide and mycophenolate mofetil. Levamisole and cyclosporine are the first alternative treatment for the steroid dependent nephrotic syndrome [56]. The workgroup of KDIGO also suggested levamisole as a corticosteroid agent and the suggested dose of levamisole is 2.5mg/kg on alternate days for at least 12 months as in most cases children suffer relapses if the dose of the levamisole is stopped suddenly [55]. Five RCTs showed that by not continued use of levamisole, the risk of relapses can be lowered to 57 percent, so it is suggested that levamisole should be taken for 12 to 24 months [2]. Levamisole is effective in reducing the relapse rates and the steroids dosage for steroid dependent nephrotic syndrome affected patients [57]. The recommended dosage of cyclosporine should be 4 to 5mg/kg/day for reducing relapses. Studies have shown that cyclosporine retains remission in almost 60 to 90 percent of children undergoing relapses after alkylating agent therapy [56].
Cyclosporine is the inhibitor which represses the immune response causing reduction in the synthesis of cytokine genes [58]. Use of cyclosporine therapy for a long time in low amount is effective but its withdrawal will lead to relapses, so treatment needs regular monitoring [59].Therapy with levamisole and cyclosporine is a long-term treatment, but have less few side effects than others [60]. Cyclophosphamide and cyclosporine are very effective in reducing relapses [61]. However, levamisole is regarded as the less costly and less toxic [62]. Table II shows different drugs along with their recommended dosage to prevent relapses.
Table II: Different drugs along their recommended dosage to prevent relapses [5, 48, 55, 56, 63]
| Drugs | Dosage | Time Period |
| 1. Prednisolone | 60mg daily,40mg on alternate days | 4 weeks |
| 2. Levamisole
|
2.5mg/kg on alternate days | 6 months |
| 3. Cyclosporine | Dosage of 2.5mg/kg | 1 year |
| 4. Cyclophosphamide | 3mg/kg/day along with 60mg/ on alternate days | 8 weeks |
| 5. Mycophenolate Mofetil | 1200 mg/ /day
|
Twice a day for 12 months |
Recent research has indicated that the monoclonal antibodies directed to B cell, like rituximab, can prove to be a better therapeutic option [64]. A case study has shown that eight doses of rituximab along with cyclosporine causes recurrence of syndrome and proteinuria in just 11 months [65]. Those patients that don’t really show response to cyclosporine A and cyclophosphamide, some alternative treatment options are available for them. Some of them are pulse methylprednisolone along with inhibitors [66]. The long-term treatment of nephrotic syndrome with prednisone can cause excessive weight gain and growth retardation [67] .
Recent Therapies
The recent therapy of immunosuppression is beneficial for immune mediated disease but not very effective in monogenic disease [68]. One of the current effective therapies is the use of anti-CD20 monoclonal antibodies [69, 70] and anti-B7-1 monoclonal antibody [71] for treatment of idiopathic nephrotic syndrome. A single-dose AAV gene therapy targeting renal podocytes has been shown to be useful in treating steroid-resistant nephrotic syndrome by restoring podocyte function [72]. Rituximab is reported as a safe first-line and effective treatment of pediatric idiopathic nephrotic syndrome, maintaining and increasing remission [73]. Transcutaneous auricular vagus nerve stimulation is reported as a well-tolerated and promising therapy for treating steroid-resistant and frequently relapsing nephrotic syndrome in children, resulting in reduced proteinuria and clinical remission, respectively [74]. It is reported that blocking the PAR-2 receptor, which regulates the passage of albumin in the kidney’s filtering units, could potentially be a treatment for kidney diseases [75]. Combination therapy with valproic acid and all-trans retinoic acid has been suggested as a potential approach for drug repurposing to improve renal function [76]. Azithromycin is reported as a new treatment for suppressing relapses in children having idiopathic nephrotic syndrome, it reduces the risk of long-term treatment [77].
CENSUS Survey Data
The Statistics Canada Census data survey from 2001-2011 for children ages 1-18 have shown a data of 711 children suffering from nephrotic syndrome for comparing the ethnic differences in long term outcomes [78]. The largest ethnic groups are shown in Figure 3. Over 10-year time period, South Asians had higher incidence of nephrotic syndrome as compared to Europeans and East Asians had smaller incidence as compared to Europeans. But the chances of relapses are higher in Europeans as compared to South Asians and East Asians. The census data survey for patients having nephrotic syndrome in various regions according to Canada Census data survey (2001-2011) is presented in Figure 3.
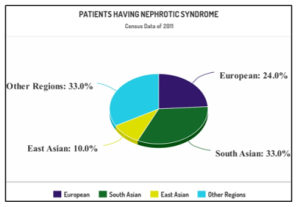
Fig. III: Incidence of Nephrotic Syndrome among children of largest ethnic groups (The statistics reported that a higher percentage; 33% of South Asian children has incidence of nephrotic syndrome while European children has 24% incidence, East Asians has 10% chances while children of other regions collectively also have 33% chances of suffering from nephrotic syndrome)
Liabilities of Corticosteroids
Therapies for patients of steroid dependent nephrotic syndrome causes many side effects which include obesity, osteoporosis, glaucoma, hypertension and psycho emotional changes [79]. Mostly levamisole does not really cause any side effect but in some cases, reversible leucopenia, seizures and liver toxicity has been observed. Use of levamisole for relapses upsets the gastrointestinal system to some extent. Cutaneous vasculitis, in which medium and small sized vessels are affected in the area of skin, is appeared in some rare cases by using levamisole [80, 81]. Cyclosporine can cause hypertension, kidney dysfunction and diabetes [82]. Children taking cyclosporine for more than 12 months are reported having tubulointerstitial lesions on kidney biopsy [83]. Corticosteroids also cause behavioral abnormalities among children. It induces aggression and attention problems [84]. Use of cyclophosphamide is a cytotoxic treatment that can result in infertility [85]. In the long-term steroid therapy, ocular complications and raised ocular pressure has been identified in many cases [86]. Due to corticosteroids therapy, patients had faced growth retardation in some of the cases [87, 88]. Main reason for growth retardation is the loss of insulin like growth factors [89]. Corticosteroids weaken the bones thus fractures can occur very easily [90]. Even the low dosage of corticosteroids causes moon face which even lasts after weeks of totally leaving corticosteroids [91]. Rituximab also causes serious complications of serum sickness which is an allergic reaction [92] and injury of lungs [93, 94]. Mycophenolate mofetil (MMF) causes blood and blood forming organs disorder [95]. The excess dosage of corticosteroids also causes Cushing syndrome which includes non-cancerous tumor formation [96].
Response of patients towards Corticosteroids in Pakistan
The patient’s response to corticosteroids treatment for nephrotic syndrome in Pakistan is presented in Table III.
Table III: Responses of nephrotic syndrome patients towards corticosteroids in Pakistan [97, 98, 99, 100, 101, 102]
| Author;
Year; Journal |
Research Study | Research Place and
Duration |
Patients | Age
Range |
Treatment with Corticosteroid | Time Period
|
Results | ||
| Male | Female | Total | |||||||
| Syed Sajid Hussain Shah and Farkhanda Hafeez; 2016;
J Ayub Med Coll Abbotabad |
Childhood Idiopathic steroid resistant nephrotic syndrome,
Different drugs and outcome |
The Children’s Hospital Lahore from Feb 2014 to May 2015 | 63 | 42 | 105 | From 1-12 Years | Tacrolimus used for 43 patients, Cyclosporine for 38 and MMF for 21 | 1 Year | 86.7% patients showedcomplete remission while partial remission in 4.7% |
| Khemani S and Moorani KN, 2016, J LiaquatUni Med Health Sci | Cyclosporine Versus Cyclophosphamide in Childhood Nephrotic Syndrome | Nephrology Unit, National Institute of Child Health, Karachi from April to September 2012 |
– |
– |
158 | From 6 months to 15 Years | Cyclosporine and Cyclophosphamide along with alternate day Prednisolone | 12 weeks | 56.3% showed complete remission while 33.5% shows partial response |
| Krishin J, Hussain M, Ayub A, and Abbasi S, 2017, JIMDC | Full dose steroid responsiveness within 8 weeks in initial treatment of childhood nephrotic syndrome | Children Hospital, Pakistan Institute of Medical Sciences, Islamabad from Jan 2012 to June 2012 | 104 | 35 | 139 | From 1 to 10 years | 60mg/ Prednisolone divided into 3 doses for 8 weeks and then switched to 40mg/ on alternate days | 8 Weeks | 24% patients’ showed response and 16% showed no response |
| Syed Sajid Hussain Shah, Naureen Akhtar and FaizaSunbleen, 2015, Rawal Medical Journal | Management of Steroid resistant Nephrotic syndrome in children with Cyclosporine – a tertiary care center experience | Children’s Hospital Lahore from March 2014 to June 2015 | 20 | 20 | 40 | From 1-12 Years | Cyclosporine with Prednisolone | 6 Month | 80% showed complete response20% showed partial response
|
| Muhammad Imran, Zulfiqar Ali, Waqas Imran Khan and Hashim Raza | Treatment Outcome in Childhood Steroid Resistant Nephrotic Syndrome with Different therapeutic Regimens | The Children’s Hospital and the Institute of Child Health, Multan from January 2006 to July 2014 | 49 | 28 | 77 | 1-15 years | Patients divided in two groups, 1st group was treated with Cyclosporine A, MMF and MP in steps while 2nd group was treated by MMF or MP | 1-5 years | In Group 1- 72% achieved complete remission while 3.4 % gave partial response and 24% gave no response, In Group 2- only 12.5% gave response |
| NazrulJaffery,Ejaz Ahmed, Muhammed Mubarak , JavedKazi and FazalAkhter, 2012,Nephrol Dial Transplant | Raised serum creatinine at presentation does not adversely affect steroid response in primary Focal Segmental Glomerulosclerosis in adults | Adult Nephrology clinic at Sindh Institute of Urology and Transplantation from January 1995 to June 2006 | 86 | 38 | 124 | >17 Years | Only 79 patients were treated with steroids | 5 Months | 50.6% patients achieved remission. Relapse occurred in 17.7%. |
Conclusion
Nephrotic syndrome, a complex renal disorder which leads to significant hyperlipidemia, edema, hypoalbuminemia and proteinuria. The primary and secondary etiological factors influence the pathogenesis of nephrotic syndrome, which is linked to alterations in the glomerular filtration barrier. This review shows that the idiopathic form, MCD (minimal change disease), is more commonly found in children, where high-dose corticosteroids are used for treatment while in adults, secondary causes such as infections and systemic diseases are more prevalent. The disease is diagnosed based on laboratory findings, the patient’s clinical presentation, and, when necessary, histopathological confirmation through renal biopsy. This study also emphasizes the two problems that patients encounter: the illness itself and the adverse effects of corticosteroid therapy. Corticosteroids, especially prednisolone, are proven to be beneficial in its treatment, which works better in steroid-sensitive patients than in steroid-resistant ones. However, there are serious hazards associated with long-term usage, such as dependency and other adverse effects. Recent clinical trials have shown that low-dose corticosteroid regimens administered on alternate days can effectively control the syndrome while reducing the risk of relapse and side effects. In order to prevent withdrawal symptoms and relapse, it is essential to reduce the dosage gradually for the best results. To maintain a balance between safety and effectiveness, future studies should concentrate on improving corticosteroid regimens and investigating alternative medicines.
Statements and Declarations
Author’s Contributions
R.N supervised and critically reviewed this study. Z. N, R.N, R.Y and G.R contributed to the conception and design of the study. The literature review and data collection were mainly performed by Z.N, M. S. R and U.I. A. R, S.A, Z. H and U.M assisted in data sorting and interpretation. All authors read and approved the final manuscript.
Funding: Nobody provided funding for this article’s publishing.
Conflicts of Interest: No conflicts of interest are disclosed by the writers.
Acknowledgement: The authors express their gratitude to everyone who
helped with their suggestions.
References
- Kalantari, S., Nafar, M., &Rezaei-Tavirani, M. (2013). Urinary proteomics in nephrotic syndrome. Journal of Paramedical Sciences, 4(4), 108-115. https://doi.org/10.22037/jps.v4i4.5007
- Lombel, R. M., Gipson, D. S., &Hodson, E. M. (2013). Treatment of steroid-sensitive nephrotic syndrome: new guidelines from KDIGO. Pediatric nephrology, 28(3), 415-426. https://doi.org/10.1007/s00467-012-2310-x
- Kakar, S., Kumar, V., & Singh, R. (2017). Latest research progress on acute nephrotic syndrome. Journal of Acute Disease, 6(6), 255. https://doi.org/10.4103/2221-6189.221288
- S. Daehn and J.S. Duffield, “The glomerular filtration barrier: a structural target for novel kidney therapies,” Nat. Rev. Drug Discovery, vol. 20, pp. 770–788, 2021. https://doi.org/10.1038/s41573-021-00242-0
- Andolino, T. P., & Reid-Adam, J. (2015). Nephrotic syndrome. Pediatrics in review, 36(3), 117-25. https://doi.org/10.1542/pir.36-3-117
- Bierzynska, A., &Saleem, M. (2017). Recent advances in understanding and treating nephrotic syndrome. F1000Research, 6. https://doi.org/10.12688/f1000research.10165.1
- Mukhi, D., Nishad, R., Menon, R. K., &Pasupulati, A. K. (2017). Novel actions of growth hormone in podocytes: implications for diabetic nephropathy. Frontiers in medicine, 4, 102. https://doi.org/10.3389/fmed.2017.00102
- Sinha, A., &Bagga, A. (2012). Nephrotic syndrome. The Indian Journal of Pediatrics, 79(8), 1045-1055. https://doi.org/10.1007/s12098-012-0776-y
- Vukojevic, K., Raguz, F., Saraga, M., Filipovic, N., Bocina, I., Kero, D., & Saraga-Babic, M. (2018). Glomeruli from patients with nephrin mutations show increased number of ciliated and poorly differentiated podocytes. Actahistochemica, 120(8), 748-756. https://doi.org/10.1016/j.acthis.2018.08.015
- Kaneko, K., Tsuji, S., Kimata, T., Kitao, T., Yamanouchi, S., & Kato, S. (2015). Pathogenesis of childhood idiopathic nephrotic syndrome: a paradigm shift from T-cells to podocytes. World Journal of Pediatrics, 11(1), 21-28. https://doi.org/10.1007/s12519-015-0003-9
- Gordillo, R., & Spitzer, A. (2009). The nephrotic syndrome. Pediatrics in review, 30(3), 94. https://doi.org/10.1542/pir.30-3-94
- Noone, D. G., Iijima, K., & Parekh, R. (2018). Idiopathic nephrotic syndrome in children. The Lancet, 392(10141), 61-74. https://doi.org/10.1016/S0140-6736(18)30536-1
- Dinçel, N., Yılmaz, E., Kaplan Bulut, İ., Hacıkara, Ş., & Mir, S. (2015). The long-term outlook to final outcome and steroid treatment results in children with idiopathic nephrotic syndrome. Renal failure, 37(8), 1267-1272. https://doi.org/10.3109/0886022x.2015.1073051
- Lane, B. M., Cason, R., Esezobor, C. I., &Gbadegesin, R. A. (2019). Genetics of childhood steroid sensitive nephrotic syndrome: an update. Frontiers in pediatrics, 7, 8. https://doi.org/10.3389/fped.2019.00008
- Çiçek, H. (2024), Introductory Chapter: An Overview of Nephritis, Nephrotic Syndrome, and Nephrosis, Novel Topics in the Diagnosis, Treatment, and Follow-Up of Nephritis, Nephrotic Syndrome, and Nephrosis. https://doi.org/10.5772/intechopen.113372.
- Hogg, R. J., Portman, R. J., Milliner, D., Lemley, K. V., Eddy, A., & Ingelfinger, J. (2000), Evaluation and management of proteinuria and nephrotic syndrome in children: recommendations from a pediatric nephrology panel established at the National Kidney Foundation conference on proteinuria, albuminuria, risk, assessment, detection, and elimination (PARADE). Pediatrics, 105(6), 1242-1249. https://doi.org/10.1542/peds.105.6.1242
- Menon, S. (2019). Acute kidney injury in nephrotic syndrome. Frontiers in pediatrics, 6, 428. https://doi.org/10.3389/fped.2018.00428
- McCaffrey, J., Lennon, R., & Webb, N. J. (2016). The non-immunosuppressive management of childhood nephrotic syndrome. Pediatric Nephrology, 31(9), 1383-1402. https://doi.org/10.1007/s00467-015-3241-0
- El Mashad, G. M., Ibrahim, S. A. E. H., & Abdelnaby, S. A. A. (2017). Immunoglobulin G and M levels in childhood nephrotic syndrome: two centers Egyptian study. Electronic physician, 9(2), 3728. https://doi.org/10.19082/3728
- Kelly, G. S., & Brady, T. M. (2016). A Child with Nephrotic Syndrome and Abdominal Pain. Clinical pediatrics, 55(7), 683-685. https://doi.org/10.1177/0009922815598861
- Hull, R. P., & Goldsmith, D. J. (2008). Nephrotic syndrome in adults. BMJ, 336(7654), 1185-1189. https://doi.org/10.1136/bmj.39576.709711.80
- Sharifi-Mood, B., Khajeh, A., Metanat, M., & Rasouli, A. (2015). Epidemiology of meningitis studied at a university Hospital in Zahedan, South-Eastern Iran. International Journal of Infection, 2(2). https://dx.doi.org/10.17795/iji-23634
- Ray, E. C., Rondon-Berrios, H., Boyd, C. R., &Kleyman, T. R. (2015). Sodium retention and volume expansion in nephrotic syndrome: implications for hypertension. Advances in chronic kidney disease, 22(3), 179-184. https://doi.org/10.1053/j.ackd.2014.11.006
- Cadnapaphornchai, M. A., Tkachenko, O., Shchekochikhin, D., & Schrier, R. W. (2014). The nephrotic syndrome: pathogenesis and treatment of edema formation and secondary complications. Pediatric nephrology, 29(7), 1159-1167. https://doi.org/10.1007/s00467-013-2567-8
- Weng, F. L., Shults, J., Herskovitz, R. M. (2005). Vitamin D insufficiency in steroid-sensitive nephrotic syndrome in remission. Pediatr Nephrol, 20, 56–63. https://doi.org/10.1007/s00467-004-1694-7
- Yang, M., Pan, X., Liang, Z., Huang, X., Duan, M., Cai, H., & Chen, L. (2019). Clinical Features of Nephrotic Syndrome with Cerebral Hemorrhage. Medical science monitor: international medical journal of experimental and clinical research, 25, 2179. https://doi.org/10.12659/msm.912466
- Candan, C., Canpolat, N., Gökalp, S., Yıldız, N., Turhan, P., Taşdemir, M., & Çalışkan, S. (2014). Subclinical cardiovascular disease and its association with risk factors in children with steroid-resistant nephrotic syndrome. Pediatric nephrology, 29(1), 95-102. https://doi.org/10.1007/s00467-013-2608-3
- Eddy, A. A., & Symons, J. M. (2003). Nephrotic syndrome in childhood. The lancet, 362(9384), 629-639. https://doi.org/10.1016/s0140-6736(03)14184-0
- Vivarelli, M., Gibson, K., Sinha, A., and Boyer, O. (2023). Childhood nephrotic syndrome. The Lancet, 402(10404), 809-824. https://doi.org/10.1016/S0140-6736(23)01051-6
- Ali, E. T. M., Makki, H. F., Abdelraheem, M. B., Makke, S. O., & Allidir, R. A. (2017). Childhood idiopathic steroid-resistant nephrotic syndrome at a Single Center in Khartoum. Saudi Journal of Kidney Diseases and Transplantation, 28(4), 851.
- Jalanko, H. (2009). Congenital nephrotic syndrome. Pediatric nephrology, 24(11), 2121-2128. https://doi.org/10.1007/s00467-007-0633-9
- Krishnan, C., Rajesh, T. V., Shashidhara, H. J., Jayakrishnan, M. P., & Geeta, M. G. (2017). Major infections in children with nephrotic syndrome. Int J ContempPediatr, 4(2), 346-50. http://dx.doi.org/10.18203/2349-3291.ijcp20170450
- Wang, Y., Bu, J., Zhang, Q., Chen, K., Zhang, J., & Bao, X. (2015). Expression pattern of aquaporins in patients with primary nephrotic syndrome with edema. Molecular medicine reports, 12(4), 5625-5632. https://doi.org/10.3892/mmr.2015.4209\
- Frățilă, V. G., Lupușoru, G., Sorohan, B. M., Obrișcă, B., Mocanu, V., Lupușoru, M., and Ismail, G. (2024). Nephrotic syndrome: From pathophysiology to novel therapeutic approaches. Biomedicines, 12(3), 569. https://doi.org/10.3390/biomedicines12030569
- Ellison, D. H. (2019). Clinical pharmacology in diuretic use. Clinical Journal of the American Society of Nephrology, 14(8), 1248-1257. https://doi.org/10.2215/cjn.09630818
- T. Christian and A.P. Maxted (2022). Optimizing the corticosteroid dose in steroid-sensitive nephrotic syndrome. Pediatr.Nephrol., 37, 37–47. https://doi.org/10.1007/s00467-021-04985-1
- Qavi, A. H., Kamal, R., &Schrier, R. W. (2015). Clinical use of diuretics in heart failure, cirrhosis, and nephrotic syndrome. International journal of nephrology. https://doi.org/10.1155/2015/975934
- Hoorn, E. J., & Ellison, D. H. (2017). Diuretic resistance. American Journal of Kidney Diseases, 69(1), 136-142. https://doi.org/10.1053/j.ajkd.2016.08.027
- Toblli, J. E., Bevione, P., Di Gennaro, F., Madalena, L., Cao, G., & Angerosa, M. (2012). Understanding the mechanisms of proteinuria: therapeutic implications. International journal of nephrology, 13. https://doi.org/10.1155/2012/546039
- Hoshino, “Renal rehabilitation: exercise intervention and nutritional support in dialysis patients,” Nutrients, vol. 13, no. 5, p. 1444, 2021. https://doi.org/10.3390/nu13051444.
- Ciriaco, M., Ventrice, P., Russo, G., Scicchitano, M., Mazzitello, G., Scicchitano, F., & Russo, E. (2013). Corticosteroid-related central nervous system side effects. Journal of pharmacology & pharmacotherapeutics, 4(1), 94. https://doi.org/10.4103/0976-500x.120975
- Deschênes, G., Dossier, C., & Hogan, J. (2019). Treating the idiopathic nephrotic syndrome: are steroids the answer? Pediatric Nephrology, 34(5), 777-785. https://doi.org/10.1007/s00467-018-3963-x
- Schijvens, A. M., ter Heine, R., De Wildt, S. N., & Schreuder, M. F. (2019). Pharmacology and pharmacogenetics of prednisone and prednisolone in patients with nephrotic syndrome. Pediatric Nephrology, 34(3), 389-403. https://doi.org/10.1007/s00467-018-3929-z
- Yuzawa, Y., Yamamoto, R., Takahashi, K., Katafuchi, R., Tomita, M., Fujigaki, Y., & Urushihara, M. (2016). Evidence-based clinical practice guidelines for IgA nephropathy 2014. Clinical and experimental nephrology, 20(4), 511-535. https://doi.org/10.1007/s10157-015-1223-y
- Kuźma-Mroczkowska, E., Skrzypczyk, P., & Pańczyk-Tomaszewska, M. (2016). Levamisole therapy in children with frequently relapsing and steroid-dependent nephrotic syndrome: a single-center experience. Central-European journal of immunology, 41(3), 243. https://doi.org/10.5114/ceji.2016.63122
- Karunamoorthy, S., Thanigachalam, D., Jeyachandran, D., Ramanathan, S., Natarajan, G., & Thoppalan, B. (2019). The safety and efficacy of mycophenolatemofetil in children and adolescents with steroid-dependent nephrotic syndrome: a single-centre study. Clinical Kidney Journal, 13(2), 179-183. https://doi.org/10.1093/ckj/sfz061
- Prasad, N., Manjunath, R., Rangaswamy, D., Jaiswal, A., Agarwal, V., Bhadauria, D., & Gupta, A. (2018). Efficacy and safety of cyclosporine versus tacrolimus in steroid and cyclophosphamide resistant nephrotic syndrome: A prospective study. Indian journal of nephrology, 28(1), 46. https://doi.org/10.4103/ijn.ijn_240_16
- Beck, L., Bomback, A. S., Choi, M. J., Holzman, L. B., Langford, C., Mariani, L. H., & Waldman, M. (2013). KDOQI US commentary on the 2012 KDIGO clinical practice guideline for glomerulonephritis. American Journal of Kidney Diseases, 62(3), 403-441. https://doi.org/10.1053/j.ajkd.2013.06.002
- Brown, E. J., Pollak, M. R., & Barua, M. (2014). Genetic testing for nephrotic syndrome and FSGS in the era of next-generation sequencing. Kidney international, 85(5), 1030-1038. https://doi.org/10.1038/ki.2014.48
- Van Husen, M., & Kemper, M. J. (2011). New therapies in steroid-sensitive and steroid-resistant idiopathic nephrotic syndrome. Pediatric nephrology, 26(6), 881-892. https://doi.org/10.1007/s00467-010-1717-5
- Mühlig, A. K., Lee, J. Y., Kemper, M. J., Kronbichler, A., Yang, J. W., Lee, J. M., & Oh, J. (2019). Levamisole in Children with Idiopathic Nephrotic Syndrome: Clinical Efficacy and Pathophysiological Aspects. Journal of clinical medicine, 8(6), 860 https://doi.org/10.3390/jcm8060860
- N. Uwaezuoke. (2015). Steroid-sensitive nephrotic syndrome in children: triggers of relapse and evolving hypotheses on pathogenesis. Ital. J. Pediatr., 41, 19. https://doi.org/10.1186/s13052-015-0123-9
- Wang, X. Q., Wang, L., Tu, Y. C., & Zhang, Y. C. (2018). Traditional Chinese medicine for refractory nephrotic syndrome: strategies and promising treatments. Evidence-Based Complementary and Alternative Medicine. https://doi.org/10.1155/2018/8746349
- Vivarelli, M., Massella, L., Ruggiero, B., & Emma, F. (2017). Minimal change disease. Clinical Journal of the American Society of Nephrology, 12(2), 332-345. https://doi.org/10.2215/cjn.05000516
- Atanda, A. T. (2012). Chapter 3: steroid-sensitive nephrotic syndrome in children. Kidney Int. Suppl, 2, 163-171. https://doi.org/10.1038/kisup.2012.16
- Alsaran, K., Mirza, K., Al-Talhi, A., & Al-Kanani, E. (2017). Experience with second line drugs in frequently relapsing and steroid dependent childhood nephrotic syndrome in a large Saudi center. International Journal of Pediatrics and Adolescent Medicine, 4(2), 66-70. https://doi.org/10.1016/j.ijpam.2017.03.002
- Samuel, E. M. K., Krishnamurthy, S., Bhanudeep, S., &Muske, S. (2017). Levamisole in frequently-relapsing and steroid-dependent nephrotic syndrome. Indian pediatrics, 54(10), 831-834. https://doi.org/10.1007/s13312-017-1144-9
- Kuga, K., Nishifuji, K., & Iwasaki, T. (2008). Cyclosporine A inhibits transcription of cytokine genes and decreases the frequencies of IL-2 producing cells in feline mononuclear cells. Journal of Veterinary Medical Science, 70(10), 1011-1016. https://doi.org/10.1292/jvms.70.1011
- Cattran, D. C., Alexopoulos, E., Heering, P., Hoyer, P. F., Johnston, A., Meyrier, A., & Praga, M. (2007). Cyclosporin in idiopathic glomerular disease associated with the nephrotic syndrome: workshop recommendations. Kidney international, 72(12), 1429-1447 https://doi.org/10.1038/sj.ki.5002553
- Kudagammana, S. T., Thalgahagoda, S., Abeyagunawardena, S., Karunadasa, U. I., & Abeyagunawardena, A. S. (2017). Efficacy of Levamisole as a single agent in maintaining remission in steroid dependant nephrotic syndrome. Sri Lanka Journal of Medicine, 26(1). http://doi.org/10.4038/sljm.v26i1.26
- Rahman, M. H., Huque, S. S., Rahman, M. A., & Uddin, G. M. (2016). Efficacy of Cyclophosphamide versus Cyclosporine in Frequent Relapse Nephrotic Syndrome–A Hospital Based Study. Journal of Pediatric Nephrology, 4(2), 60-64. https://doi.org/10.22037/jpn.v4i2.11716
- Gruppen, M. P., Bouts, A. H., Jansen-van der Weide, M. C., Merkus, M. P., Zurowska, A., Maternik, M., & Schurmans, T. (2018). A randomized clinical trial indicates that levamisole increases the time to relapse in children with steroid-sensitive idiopathic nephrotic syndrome. Kidney international, 93(2), 510-518. https://doi.org/10.1016/j.kint.2017.08.011
- Gao, C. L., & Xia, Z. K. (2017). Interpretation of guidelines for the diagnosis and treatment of steroid-resistant nephrotic syndrome. Chinese journal of pediatrics, 55(11), 810-812. https://doi.org/10.3760/cma.j.issn.0578-1310.2017.11.003
- Kallash, M., Smoyer, W. E., & Mahan, J. D. (2019). Rituximab use in the management of childhood nephrotic syndrome. Frontiers in pediatrics, 7,178. https://doi.org/10.3389/fped.2019.00178
- Fujinaga, S., & Sakuraya, K. (2017). Repeated administrations of rituximab along with steroids and immunosuppressive agents in refractory steroid-resistant nephrotic syndrome. Indian Pediatr, 54, 49-50. https://doi.org/10.1007/s13312-017-0996-3
- Mori, K., Honda, M., & Ikeda, M. (2004). Efficacy of methylprednisolone pulse therapy in steroid-resistant nephrotic syndrome. Pediatric Nephrology, 19(11), 1232-1236. https://doi.org/10.1007/s00467-004-1584-z
- Ehren, R., Benz, M. R., Doetsch, J., Fichtner, A., Gellermann, J., Haffner, D., & Konrad, M. (2018). Initial treatment of steroid-sensitive idiopathic nephrotic syndrome in children with mycophenolatemofetil versus prednisone: protocol for a randomised, controlled, multicentre trial (INTENT study). BMJ open, 8(10). https://doi.org/10.1136/bmjopen-2018-024882
- Saleem, M. A. (2019). Molecular stratification of idiopathic nephrotic syndrome. Nature Reviews Nephrology, 5(12), 750-765. https://doi.org/10.1038/s41581-019-0217-5
- Ravani, P., Ponticelli, A., Siciliano, C., Fornoni, A., Magnasco, A., Sica, F., & Ghio, L. (2013). Rituximab is a safe and effective long-term treatment for children with steroid and calcineurin inhibitor–dependent idiopathic nephrotic syndrome. Kidney international, 84(5), 1025-1033. https://doi.org/10.1038/ki.2013.211
- Bonanni, A., Calatroni, M., D’Alessandro, M., Signa, S., Bertelli, E., Cioni, M., & Bertelli, R. (2018). Adverse events linked with the use of chimeric and humanized anti‐CD20 antibodies in children with idiopathic nephrotic syndrome. British journal of clinical pharmacology, 84(6), 1238-1249. https://doi.org/10.1111/bcp.13548
- Yu, C. C., Fornoni, A., Weins, A., Hakroush, S., Maiguel, D., Sageshima, J., & Campbell, K. N. (2013). Abatacept in B7-1–positive proteinuric kidney disease. New England Journal of Medicine, 369(25), 2416-2423. https://doi.org/10.1056/nejmoa1304572
- Y. Ding, V. Kuzmuk, S. Hunter, A. Lay, B. Hayes, M. Beesley, M. A. Saleem. (2023). Adeno-associated virus gene therapy prevents progression of kidney disease in genetic models of nephrotic syndrome. Science Translational Medicine, 15(708), 8226. https://doi.org/10.1126/scitranslmed.abc8226.
- Zhang, Y. Jin, F. Liu, Q. Li, Y. Xie, G. Huang, J. Chen, X. He, S. He, H. Fu, J. Wang, H. Shen, J. Mao. (2024). Rituximab as a first-line therapy in children with new-onset idiopathic nephrotic syndrome. Clinical Kidney Journal, 348. https://doi.org/10.1093/ckj/sfae348.
- Merchant, S. Zanos, T. Datta-Chaudhuri. (2022). Transcutaneous auricular vagus nerve stimulation (taVNS) for the treatment of pediatric nephrotic syndrome: a pilot study. Bioelectron Med, 8(1). https://doi.org/10.1186/s42234-021-00084-6.
- Sharma, V. Prasad, E. T. McCarthy. (2007). Chymase increases glomerular albumin permeability via protease-activated receptor-2. Mol. Cell Biochem., 297, 161–169. https://doi.org/10.1007/s11010-006-9342-0.
- Varghese and A. Majumdar. (2024). All-trans-retinoic acid and valproic acid: A combinatorial approach for the treatment of nephrotic syndrome. Current Research in Biotechnology, 7, 100181. https://doi.org/10.1016/j.crbiot.2024.100181.
- Hara, H., & Hirano, D. (2018). Azithromycin suppressed relapses of idiopathic nephrotic syndrome in a child. Clinical kidney journal, 11(1), 54-55. https://doi.org/10.1093/ckj/sfx099
- Banh, T. H., Hussain-Shamsy, N., Patel, V., Vasilevska-Ristovska, J., Borges, K., Sibbald, C., & Reddon, M. (2016). Ethnic differences in incidence and outcomes of childhood nephrotic syndrome. Clinical Journal of the American Society of Nephrology, 11(10), 1760-1768. https://doi.org/10.2215/cjn.00380116
- Hahn, D., Hodson, E. M., Willis, N. S., & Craig, J. C. (2020). Corticosteroid therapy for nephrotic syndrome in children. Cochrane Database of Systematic Reviews, (3). https://doi.org/10.1002/14651858.CD001533.pub6
- Bagga, A. (2008). Revised guidelines for management of steroid-sensitive nephrotic syndrome. Indian journal of nephrology, 18(1), 31. https://doi.org/10.4103/0971-4065.41289
- Youssef, D. M., Al-atif, A. M. A., El-Khateeb, S. S. H., & Elshal, A. S. (2018). Evaluation of interleukin-18 in children with steroid-sensitive nephrotic syndrome before and after using levamisole. Saudi Journal of Kidney Diseases and Transplantation, 29(3), 591. https://doi.org/10.4103/1319-2442.235173
- Yu, X., Ruan, L., Qu, Z., Cui, Z., Zhang, Y., Wang, X., & Liu, G. (2017). Low-dose cyclosporine in treatment of membranous nephropathy with nephrotic syndrome: effectiveness and renal safety. Renal failure, 39(1), 688-697. https://doi.org/10.1080/0886022x.2017.1373130
- Iijima, K., Hamahira, K., Tanaka, R., Kobayashi, A., Nozu, K., Nakamura, H., & Yoshikawa, N. (2002). Risk factors for cyclosporine-induced tubulointerstitial lesions in children with minimal change nephrotic syndrome. Kidney international, 61(5), 1801-1805. https://doi.org/10.1046/j.1523-1755.2002.00303.x
- Mishra, O. P., Basu, B., Upadhyay, S. K., Prasad, R., & Schaefer, F. (2010). Behavioural abnormalities in children with nephrotic syndrome. Nephrology Dialysis Transplantation, 25(8), 2537-2541. https://doi.org/10.1093/ndt/gfq097
- Al Salloum, A. A., Muthanna, A., Bassrawi, R., Al Shehab, A. A., Al Ibrahim, A., Islam, M. Z., & Alhasan, K. (2012). Long-term outcome of the difficult nephrotic syndrome in children. Saudi Journal of Kidney Diseases and Transplantation, 23(5), 965. https://doi.org/10.4103/1319-2442.100877
- Gaur, S., Joseph, M., Nityanandam, S., Subramanian, S., Koshy, A. S., Vasudevan, A., & Iyengar, A. (2014). Ocular complications in children with nephrotic syndrome on long term oral steroids. The Indian Journal of Pediatrics, 81(7), 680-683. https://doi.org/10.1007/s12098-014-1338-2
- Leroy, V., Baudouin, V., Alberti, C., Guest, G., Niaudet, P., Loirat, C., & Simon, D. (2009). Growth in boys with idiopathic nephrotic syndrome on long-term cyclosporin and steroid treatment. Pediatric Nephrology, 24(12), 2393-2400. https://doi.org/10.1007/s00467-009-1266-y
- Donatti, T. L., Koch, V. H. K., Takayama, L., & Pereira, R. M. R. (2011). Effects of glucocorticoids on growth and bone mineralization. Jornal de pediatria, 87(1), 4-12. https://doi.org/10.2223/jped.2052
- Park, S. J., & Shin, J. I. (2011). Complications of nephrotic syndrome. Korean journal of pediatrics, 54(8), 322-328. https://doi.org/10.3345/kjp.2011.54.8.322
- Klein, G. L. (2015). The effect of glucocorticoids on bone and muscle. Osteoporosis and sarcopenia, 1(1), 39-45. https://doi.org/10.1016/j.afos.2015.07.008
- Oray, M., Abu Samra, K., Ebrahimiadib, N., Meese, H., & Foster, C. S. (2016). Long-term side effects of glucocorticoids. Expert opinion on drug safety, 15(4), 457-465. https://doi.org/10.1517/14740338.2016.1140743
- Maeda, R., Kawasaki, Y., Ohara, S., Suyama, K., &Hosoya, M. (2018). Serum sickness with refractory nephrotic syndrome following treatment with rituximab. CEN case reports, 7(1), 69-72. https://doi.org/10.1007/s13730-017-0297-7
- Grenda, R., Jarmużek, W., Rubik, J., Migdał, M., &Pronicki, M. (2015). Fatal rituximab‐associated lung injury syndrome in a patient treated with rituximab for recurrence of post‐transplant nephrotic syndrome. Pediatric transplantation, 19(5), 115-120. https://doi.org/10.1111/petr.12481
- Spatafora, M., Bellini, T., Giordano, C., & Ghiggeri, G. M. (2015). A mild form of rituximab-associated lung injury in two adolescents treated for nephrotic syndrome. Case Reports. https://doi.org/10.1136/bcr-2015-212694
- Kopač, M. (2018). Nephrotic Syndrome in Children–Present State and Future Perspectives. Journal of Nephrology Research, 4(1), 139-145. http://dx.doi.org/10.17554/j.issn.2410-0579.2018.04.28
- Nantarakchaikul, P., & Nimdet, K. (2015). An infantile exogenous Cushing syndrome caused by topical steroid. International journal of pediatric endocrinology, 2015(1), 45. https://doi.org/10.1186/1687-9856-2015-s1-p45
- Ehren, R., Benz, M. R., Brinkkötter, P. T. (2021). Pediatric idiopathic steroid-sensitive nephrotic syndrome: diagnosis and therapy — short version of the updated German best practice guideline (S2e) — AWMF register no. 166-001, 6/2020. Pediatr Nephrol, 36, 2971–2985. https://doi.org/10.1007/s00467-021-05135-3.
- S. Dhooria, S. Bhargava, D. Bhat. (2024). Efficacy and safety of oral cyclophosphamide versus mycophenolate mofetil in childhood nephrotic syndrome: an open-label comparative study. BMC Nephrol, 25, 296. https://doi.org/10.1186/s12882-024-03739-z.
- M. Schijvens, N. Teeninga, E. M. Dorresteijn. (2021). Steroid treatment for the first episode of childhood nephrotic syndrome: comparison of the 8- and 12-weeks regimen using an individual patient data meta-analysis. Eur. J. Pediatr., 180, 2849–2859. https://doi.org/10.1007/s00431-021-04035-w.
- A. W. Kashif, A. Ranjan, S. Kalra, U. D. S. Uttpal, and A. Malik. (2022). Clinicopathological spectrum and treatment outcome of idiopathic steroid-resistant nephrotic syndrome in children at a tertiary care center. Med. J. Armed Forces India, 78(3), 291-295. https://doi.org/10.1016/j.mjafi.2020.11.004.
- Welegerima, M. Feyissa, and T. Nedi. (2021). Treatment outcomes of pediatric nephrotic syndrome patients treated in Ayder Comprehensive Specialized and Mekelle General Hospitals, Ethiopia. Int. J. Nephrol. Renovascular Dis., 14, 149–156. https://doi.org/10.2147/IJNRD.S310567
- Jafry, N., Ahmed, E., Mubarak, M., Kazi, J., & Akhter, F. (2012). Raised serum creatinine at presentation does not adversely affect steroid response in primary focal segmental glomerulosclerosis in adults. Nephrology Dialysis Transplantation, 27(3), 1101-1106. https://doi.org/10.1093/ndt/gfr430