FAHEEM NAWAZ1, AMIR NADEEM*1, ABDUL QAYYUM KHAN SULEHRIA1, ALTAF HUSSAIN1, NAVEED AKHTAR1, SARA HAYEE1, SHAISTA SHABIR2
1Department of Zoology, Government College University, Lahore, Pakistan
2Institute of Zoology, University of the Punjab, Lahore, Pakistan
Received: 14-Jan-2023 / Revised and Accepted: 15-Jan-2023 / Published On-Line: 21-Jan-2023
https://doi.org/10.56819/pjest.v3i2.82
ABSTRACT:
Zooplanktons are a very important part of a water reservoir. They are a link between producers and consumers. Although there are many types of zooplanktons, copepods are very important as they are bio-indicator of the eutrophic condition of a water reservoir. The present study studied the spatio-temporal dispersal of copepods at Jallo Lake Wildlife Park, Lahore. A total of ten species belonging to seven orders, including nauplius larvae, were identified. The physicochemical parameters were observed, i.e., water and air temperature, oxygen saturation, pH, transparency, dissolved oxygen and conductivity. ANOVA was applied to all the physicochemical parameters. It was highly significant for all parameters (P= 0.000, α = 0.05) except for the transparency. Copepod diversity and density were at their peak in June. Pearson correlation was applied between the physicochemical parameters and copepods. Air temperature, water temperature, and conductivity were positively correlated, whereas dissolved oxygen, oxygen saturation, transparency, and pH were negatively correlated. The quantity of the Shannon Weaver index and Simpson index was studied and calculated every month, showing fluctuations during the study period. The results revealed that Diacyclops thomasi and Nauplius larvae were the most abundant species.
Keywords: Copepod, Shannon Weaver Index, Simpson Index, Physico-Chemicals, Pearson Correlation.
Introduction:
The class name ‘Copepod’ has been taken from two Greek words, ‘Kope’ meaning ‘Oar and poda’ meaning – foot or pedal [1]. Copepods show massive diversity due to their ability to exist and survive in various habitats, including freshwater and marine [2]. It is said that copepod species
are linked to all animal phyla due to their ability to recycle organic matter. Most of these species are 1-2 mm, i.e., 0.04 inch to 0.08 inch [3].
The general body of a copepod is divided into two major parts. The Anterior region is called “Prosome,” while the posterior region is named “Urosome.” A flexing point in the body connects these two regions. Different copepods have variations in the number of segments in the prosomes. As for external morphology, almost all copepods have a unique body shape that appears like a teardrop, generally with large antennae. Copepods possess two pairs of antennae; the first one is often longer and conspicuous. The first antenna is modified according to the function, mainly feeding. Copepods use smaller organisms as their prey [4].
Being members of Kingdom Animalia, Copepods belong to Phylum Arthropoda, Subphylum Crustacea, and Subclass Copepoda. The subclass Copepoda is further divided into ten orders, i.e., Calanoida, Cyclopoida, Gelyelloida, Harpacticoida, Misaphrioida, Monsteillioida, Mornomilloida, Platycopioida, Poecilostomotia, Siphonostomaloida. Thirteen thousand copepod species were designated, and approximately some of them are parasitic [1].
Copepods have variations in their speeds. One type of locomotion is slow and steady, carried out with mouthparts or anterior portions. The other is consecutive jumping with small gaps. This type of movement is done with the appendages attached to the thorax. The swimming speed of the copepods was influenced by the increase in food concentration and filtration rate of food [5-6]. Copepods are primary consumers of phytoplanktons and are a food source for larval and juvenile fish [7]. Many studies have revealed that physico-chemical parameters like pH, transparency and temperature were responsible for this distribution of zooplanktons [8]. The above literature shows the importance of copepods in shaping any water reservoir’s overall community. Moreover, the abundance and diversity of these organisms are useful in qualifying and monitoring the water quality of any water reservoir.
OBJECTIVES OF THE STUDY
The following are the major objectives of the present study;
- To collect and identify copepods in the Jallo lake to study their distribution and abundance.
- To find out the Physicochemical limitations of the water and its interpretations on the copepod’s population.
- To quantify the copepod population in various months by diversity indices, species richness, and evenness.
MATERIALS AND METHODS
The following materials and methods were used during this study;
Sampling Region
An artificial lake situated in Jallo Wildlife Park was selected for sampling. It is commonly called Jallo Park, developed in 1978 in the Lahore District. It is situated at a distance of 28 kilometres East of Lahore city, towards ―Wagha Border having an area of 456 acres. The study site is at 31.571926 and 74.469024 latitude and longitude, respectively.
The Jallo lake is a circular lake. Four spots were selected and named, e.g., the Eastern side, JS1; the Southern side, JS2; the Western side JS3; and the Northern side, JS4 starting from the boating point or boat stand. Each spot was divided into three sub-spots named a, b, and c [9] (Figure 1).
Water Sampling
Water sampling continued for the whole year from October 2012 to August 2013 every month. The samples were taken in the last week of every month between 10:00 am to 01:00 pm. Water samples were taken in BOD bottles of 1-litre capacity. Sampling containers were drenched in HCl acid, washed with sanitized water, and dried. They were bathed with the lake water two or three times to acclimatize the bottles before sample collection. All the physicochemical parameters were measured with their respective meters.
Identification and Counting of Copepods
Copepods were identified with the help of suitable keys based on their external morphology [10]. Sedgwick Rafter and an inverted Olympus microscope were used to analyze copepods quantitatively. Copepods were photographed with a camera (5.0- Megapixel Cannon) fitted on a microscope for a visual recording. Sedgwick Rafter is a slide with a carrying capacity of 1 ml water sample. It is 50 mm long and 20mm wide. A complete row along the length is called a ―strip, so there are 20 rows in a Sedgwick Rafter.
Number/ml = C x 1000 mm / L x D x W x S

Fig. 1: Jallo Lake – Aerial view. Courtesy: Google Maps
* Eastern side, JS1; the Southern side, JS2; the Western side JS3; and the Northern side, JS4.
Estimation of Population Density and Diversity of Copepods
The two indices were used to find the biodiversity.
It was by Shannon-Weaver equation as given below
H= – ∑ Pi (lnPi) ………………….[11]
D = ∑ n (n-1) / N (N-1) ………………….[12]
This equation, called Simpson’s index of dominance, is utilized to find out how the individuals of two species are closely related. Similarly, Simpson’s index of diversity (SID), Simpson’s reciprocal index (SRI), Simpson’s index of diversity (SID), Simpson’s reciprocal index (SRI), Species richness (SR), SR = (s – 1)/log n [13], Species evenness or equitability (E), E = H/logn s [14-15]. (Table 01).
Statistical Analysis
Analysis of variance (ANOVA) was applied to the given data of copepods obtained in different months from different sampling sites to determine their relative difference. Pearson’s correlation test was applied to determine the relationships between the observed environmental parameters and copepod species. The software used for ANOVA and Pearson’s correlation was Minitab 13. The graphs were plotted in Microsoft Excel 2010.
Results and Discussion:
The following results were made based on collected data;
Physico-Chemical Parameters
Air Temperature, as well as water temperature, ranged from 13.97°C to 35.2°C and 16.32°C ± 0.085°C to 39.6°C ±0.040°C respectively. The maximum value was calculated in June, and the lowest was in December. Temperature positively correlated with copepod density. Similar findings were reported by many other researchers [16-19]. Dissolved oxygen was highest in December (10.7 mg/L) and lowest in June (6.7 mg/L). In October and June, water conductivity varied between 315.1µS/sec and 513.0µS/sec, respectively. Two research groups, one in Kashmir and the other in Brazil, also studied the zooplankton quantity, i.e., Rotifers, Cladocerans, and Copepods, and found a positive correlation with electrical conductivity [20-21] (Graph 1).
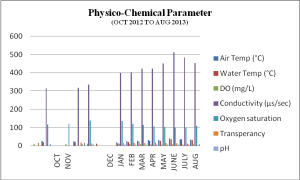
Graph 1: Physico-Chemical Parameter (Oct 2012 To Aug 2013)
Oxygen Saturation ranged between 100.3 and 119.8 in June and February, respectively. A negative correlation exists between copepods and dissolved oxygen. A previous study reported similar results [22] (Graph 01). Transparency was found to behave elevated peak in October (16 cm) and lowermost in June (10.9 cm). The least changes were seen in the pH value; it showed a variation from 8.3 (October) to 7.4 (March). It has been reported earlier that copepods and pH were a periodical correlation throughout the year [23]. The Mesocyclops species were found in March and April, and a similar observation was recorded earlier (Table 1) [24-25].
Density and Diversity of Copepods
The population density of each species was calculated each month. The mean and total sum were calculated from data obtained monthly basis. The rich diversity was detected in May, June, and July, but it was highest in June. Ten species belonging to 07 orders and Nauplius Larvae were observed (Table 1). After identification, each species was photographed to keep a pictorial record (Figure 2). The total population density of each species was calculated for each month with mean and sum values tabulated. The least copepod species were found in November. Diacyclops bicuspidatus (0.3125), Diacyclops thomasi (0.58523), Microcyclops varicans (0.31818), Mesocyclops species (0.38636), and Nauplius were almost found in all months. Still, they quantitatively showed variation; however, Eucyclops phaleratus, Eucalanus attenuates, Eucyclops aglis, Eucyclops elegans, and Ergaslius species were found from April to August. The dominant and most abundant species of copepods were D. thomasi (0.58523) and Nauplius (0.67043), and were present at all study sites [26]. Eucalanus attenutus was the least common among all species (0.07955) (Figure 1).
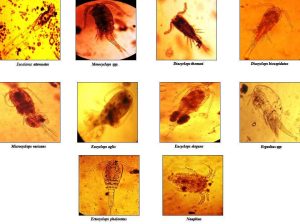
Fig. 2: Ten species of Copepods identified from the study area.
Table 1. POPULATION DENSITY OF COPEPODS SPECIES IDENTIFIED FROM JALLO LAKE LAHORE (OCT-2012 TO AUG 2013)
| Species | October | November | December | January | February | March | April | May | June | July | August | MEAN | SUM |
| Diacyclops bicuspidatus | 0.4375 | 0.3125 | 0.25 | 0.125 | 0.125 | 0.1875 | 0.3125 | 0.5 | 0.9375 | 0.125 | 0.125 | 0.3125 | 3.4375 |
| Diacyclops thomasi | 0.4375 | 0.3125 | 0.25 | 0.5 | 0.375 | 0.3125 | 0.625 | 0.9375 | 1.1875 | 0.8125 | 0.6875 | 0.58523 | 6.4375 |
| Ectocyclops phaleratus | 0 | 0 | 0 | 0 | 0 | 0.125 | 0.25 | 0.1875 | 0.1875 | 0.625 | 0 | 0.125 | 1.375 |
| Ergaslius spp. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.625 | 0.0625 | 0.0625 | 0.06818 | 0.75 |
| Eucalanus attenuates | 0 | 0 | 0 | 0 | 0 | 0 | 0.25 | 0 | 0.25 | 0.375 | 0 | 0.07955 | 0.875 |
| Eucyclops aglis | 0 | 0 | 0 | 0 | 0.5 | 0 | 1 | 1.125 | 1.1875 | 1.0625 | 0.625 | 0.5 | 5.5 |
| Eucyclops elegans | 0 | 0 | 0 | 0 | 0 | 0 | 0.25 | 0.25 | 0 | 0.125 | 0 | 0.05682 | 0.625 |
| Microcyclops varicans | 0.3125 | 0 | 0.375 | 0.25 | 0 | 0.125 | 0.1875 | 0.25 | 0.6875 | 0.9375 | 0.375 | 0.31818 | 3.5 |
| Mesocyclops spp. | 0.0625 | 0.25 | 0.25 | 0.5625 | 0.4375 | 0.125 | 0 | 0.5625 | 1.0625 | 0.8125 | 0.125 | 0.38636 | 4.25 |
| Nauplius | 0.25 | 0.4372 | 0.5 | 0.8125 | 0.375 | 0.5625 | 0.625 | 1.25 | 1.0625 | 0.5625 | 0.9375 | 0.67043 | 7.3747 |
| TOTAL | 1.5 | 1.3122 | 1.625 | 2.25 | 1.8125 | 1.4375 | 3.5 | 5.0625 | 7.1875 | 5.5 | 2.9375 | 3.10225 | 34.1247 |
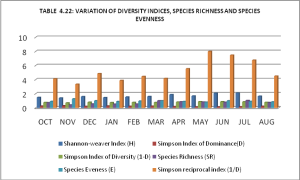
The values of the Shannon Weaver index were studied and calculated every month, which ranged from 1.39936 to 2.1050. Simpson’s index of dominance (D) was at its peak in October (0.2431) and November (0.2992), and then it dropped down to 0.1249 in May. The Simpson Diversity Index values fluctuated between 0.7008-0.875. It was found to be maximum in May and minimum in November. Species richness was observed maximum in October (7.3132) and lowermost November (0.4079). It was observed that species evenness remained constant throughout the study from October to August. The maximum value (1.2737) of species evenness was found in November, and the minimum value (0.8039) of species evenness was found in May [27].
The value of the Simpson Reciprocal Index (3.3422) was minimum in November and maximum in May (8.001) and June (7.4675). The main reason for the presence of copepods in large numbers was the easily available food in the present lake. Moreover, the stagnant water was suitable for the algae, which was essential in establishing the food web [14]. The increased population density of copepods may be due to the better ecological conditions at Jallo Lake. Organic matter was the main reason for producing copepods at a high level in a stagnant water body [28-30] (Graph 2).
THE FOLLOWING TABLES (TABLE 2 to 8) SHOWING THE ANALYSIS OF VARIANCES OF DIFFERENT PHYSICO-CHEMICAL PARAMETERS
TABLE 2
ANOVA OF WATER TEMPERATURE
| Source | DF | SS | MS | F | P |
| Months | 10 | 2494.095 | 249.410 | 1761.48 | 0.000 |
| Error | 33 | 4.672 | 0.142 | ||
| Total | 43 | 2498.768 |
TABLE 3
ANOVA OF AIR TEMPERATURE
| Source | DF | SS | MS | F | P |
| Months | 10 | 2303.83 | 230.38 | 32.59 | 0.000 |
| Error | 33 | 233.27 | 7.07 | ||
| Total | 43 | 2537.10 |
TABLE 4
ANOVA OF pH
| Source | DF | SS | MS | F | P |
| Months | 10 | 5.14500 | 0.51450 | 53.90 | 0.000 |
| Error | 33 | 0.31500 | 0.00955 | ||
| Total | 43 | 5.46000 |
TABLE 5
ANOVA OF OXYGEN SATURATION
| Source | DF | SS | MS | F | P |
| Months | 10 | 6730.336 | 673.034 | 3503.17 | 0.000 |
| Error | 33 | 6.340 | 0.192 | ||
| Total | 43 | 6736.676 |
TABLE 6
ANOVA OF ELECTRICAL CONDUCTIVITY
| Source | DF | SS | MS | F | P |
| Months | 10 | 174472.4 | 17447.2 | 6.2E+05 | 0.000 |
| Error | 33 | 0.9 | 0.00 | ||
| Total | 43 | 174473.3 |
TABLE 7
ANOVA OF DISSOLVED OXYGEN
| Source | DF | SS | MS | F | P |
| Months | 10 | 69.5868 | 6.9587 | 169.47 | 0.000 |
| Error | 33 | 1.3550 | 0.0411 | ||
| Total | 43 | 70.9418 |
TABLE 8
ANOVA OF TRANSPARENCY
| Source | DF | SS | MS | F | P |
| Months | 10 | 176.1164 | 17.6116 | * | * |
| Error | 33 | 0.0000 | 0.0000 | ||
| Total | 43 | 176.1164 |
Note: From Tables 2 to 8, DF stands for Degree of Freedom; SS stands for Sum of Squares; MS stands for Mean of Squares; F stands for f-Distribution, and P stands for Probability
Analysis of variance (ANOVA) was applied to study the significant differences in all parameters. The difference in physicochemical parameters was statistically significant (P= 0.000) for all parameters except for the transparency, which showed an infinite value exceeding the range (Tables 2 to 8). Pearson Correlation was applied to study the effect of physicochemical parameters on copepods. It showed that air temperature (AT), water temperature (WT), and electrical conductivity (C) were positively correlated with the copepod’s diversity, which means that with the increase in parameter values, the density and diversity of copepods increased. At the same time, pH, transparency (T), dissolved oxygen (DO), as well as oxygen saturation (OS) have a negative correlation with copepods (Table 9).
TABLE 9
| Copepods | AT | WT | DO | C | OS | T | |
| AT | 0.835
0.001 |
||||||
| WT | 0.729
0.011 |
0.952
0.000 |
|||||
| DO | -0.759
0.007 |
-0.952
0.000 |
-0.896
0.000 |
||||
| C | 0.840
0.001 |
0.801
0.003 |
0.747
0.008 |
-0.713
0.014 |
|||
| OS | -0.725
0.012 |
-0.933
0.000 |
-0.884
0.000 |
0.998
0.000 |
-0.686
0.020 |
||
| T | -0.401
0.221 |
-0.384
0.244 |
-0.270
0.422 |
0.185
0.585 |
-0.557
0.075 |
0.143
0.674 |
|
| pH | -0.471
0.144 |
-0.306
0.360 |
-0.287
0.000 |
0.327
0.327 |
-0.732
0.010 |
0.321
0.335 |
0.117
0.733 |
Pearson Correlation values
Note: AT (Air Temperature); WT (Water Temperature); DO (Dissolved Oxygen); C (Conductivity); OS (Oxygen Saturation); T (Turbidity).
Conclusion:
It is concluded from the above-cited study that the temporal and spatial quantity of copepods is affected by the proportion to the physicochemical parameters. The temperature of air and water, pH, conductivity and dissolved oxygen are necessary to support life in a water ecosystem. These parameters affect the presence of zooplanktons like copepods. All the physicochemical parameters were positively correlated with copepods supporting their growth and promoting their development. This point gets support from the abundant presence of copepod larvae in the lake. The water reservoir studied is an artificial lake used for recreation. It involves boating by the general public. Lake water was less affected by the purpose mentioned above. It also shows that water replacement was good enough to minimize eutrophication. Overall, a moderate diversity and density of copepods were found. Better trophic conditions of the lake helped the copepods to survive and reproduce. Copepods and their larvae were involved in small food chains, leading to a food web. So, water reservoirs with good trophic conditions can support animal life like zooplanktons which are primary consumers and transfer energy from producers to secondary consumers.
Drawbacks:
The precision of results can be improved by studying at the DNA level. Due to the non-availability of funds, Genetic-level analysis was not conducted.
Author’s Contribution: Abdul Qayyum Khan Sulehria: Conceived the idea, designed the simulated work and supervised the whole study project. Faheem Nawaz: Did the sample collection data acquisition, executed simulated work and literature review, and wrote the basic draft of the manuscript. Altaf Hussain: Helped in the identification, analysis and interpretation of data. Amir Nadeem: Critically revised the manuscript, made the language and grammatical edits and did all the correspondence. Naveed Akhtar: Helped in sample collection and shifting to the research lab from the collection site. Sara Hayee: Did statistical analysis. Shahista Shabir: Did the Photography.
Funding: The publication of this article was funded by no one.
Conflicts of Interest: The authors declare no conflict of interest.
Acknowledgement: The authors would like to thank Dr. Nusrat Jahan, The Chairperson, Department of Zoology, GC University Lahore-Pakistan, for providing all the possible facilities during this research.
REFERENCES:
[1] S. A. Miller and J. P. Harley, Zoology. McGraw-Hill Inc. New York. USA, 2007.
[2] G. G. Marten and J. W. Reid, “Cyclopoid copepods,” Journal of the American Mosquito Control Association, vol. 23, no. sp2, pp. 65-92, 2007.
[3] W. T. Edmondson, H. B. Ward, and G. C. Whipple, Freshwater Biology, 2nd ed. John Wiley and Sons. New York and London, 1959.
[4] K. Roche, “Prey features affecting ingestion rates by Acanthocyclops robustus (Copepoda: Cyclopoida) on zooplankton,” Oecologia, vol. 83, no. 1, pp. 76-82, 1990.
[5] L. A. Van Duren and J. J. Videler, “Swimming behaviour of developmental stages of the calanoid copepod Temora longicornis at different food concentrations,” Marine Ecology Progress Series, vol. 126, pp. 153-161, 1995.
[6] R. K. Lam and B. W. Frost, “Model of copepod filtering response to changes in size and concentration of food,” Limnology and Oceanography, vol. 21, no. 4, pp. 490-500, 1976.
[7] G. A. Boxshall and S. H. Halsey, An introduction to copepod diversity. Ray Society, Andover, UK, 2004, p. xv + vii + 966 pp.
[8] O. O. Stephen, M. Onimisi, and O. Martins, “The zooplankton of Ojofu Lake in Anyigba, Dekina LGA, Kogi State, Nigeria,” Researchers World, vol. 2, no. 2, pp. 114-122, 2011.
[9] R. G. D. Steel and J. H. Torrie, Principles and procedures of statistics, a biometrical approach (no. Ed. 2). McGraw-Hill Kogakusha, Ltd. Tokyo, Japan, 1980, p. 633.
[10] J. Huggett and J. Bradford-Grieve, “Guide to some common copepods in the Benguela Current LME,” in Zooplankton Workshop, Swakopmund, Namibia, 1944.
[11] C. E. Shannon and W. Weaver, The mathematical theory of communication. University of Illinois Press, Urbana., 1949, p. 125.
[12] E. H. Simpson, “Measurement of Diversity,” Nature, vol. 163, p. 688, 1949.
[13] R. Margalef, Perspectives in Ecological Theory. University of Chicago Press. Chicago, USA., 1968.
[14] E. C. Pielou, “The measurement of diversity in different types of biological collections,” Journal of Theoretical Biology, vol. 13, pp. 131-144, 1966.
[15] R. K. Peet, “The measurement of species diversity,” Annual Review of Ecology and Systematics, vol. 5, pp. 285-307, 1974.
[16] P. S. Joshi, “Studies on zooplanktons of Rajura lake of Buldhana district, Maharashtra India,” Science Research Reporter, vol. 1, no. 3, pp. 132-137, 2011.
[17] B. Salve and C. Hiware, “Zooplankton diversity of wan reservoir, Nagpur (MS) India,” Trends Research in science and Technology, vol. 2, no. 1, pp. 39-48, 2010.
[18] K. B. Koli and D. V. Muley, “Study of zooplankton diversity and seasonal variation with special reference to physicochemical parameters in Tulshi Reservoir of Kolhapur District (MS), India,” E-International Scientific Research Journal, vol. 4, no. 1, pp. 38-46, 2012.
[19] L.-C. Tseng, R. Kumar, H.-U. Dahms, Q.-C. Chen, and J.-S. Hwang, “Monsoon-driven succession of copepod assemblages in coastal waters of the northeastern Taiwan Strait,” Zoological Studies, vol. 47, no. 1, pp. 46-60, 2008.
[20] I. Ahangar, D. Saksena, and M. Mir, “Seasonal Variation in Zooplankton Community Structure of Anchar lake, Kashmir,” Universal Journal of Environmental Research Technology, vol. 2, no. 4, 2012.
[21] G. Perbiche-Neves, C. Fileto, J. Laço-Portinho, A. Troguer, and M. Serafim-Júnior, “Relations among planktonic rotifers, cyclopoid copepods, and water quality in two Brazilian reservoirs,” Latin American Journal of Aquatic Research, vol. 41, no. 1, pp. 138-149, 2013.
[22] U. Roy, B. K. Shaha, K. Mazhabuddin, F. Haque, and G. Sarower, “Study on the diversity and seasonal variation of zooplankton in a brood pond, Bangladesh,” Marine. res. aqua., vol. 1, no. 1, pp. 30-37, 2010.
[23] S. Sharma et al., “Population dynamics and seasonal abundance of zooplankton community in Narmada River (India),” Researcher, vol. 2, no. 9, pp. 1-9, 2010.
[24] A. K. Singh, “Quality assessment of surface and subsurface water of Damodar river basin,” Indian Journal of Environmental Health, vol. 44, no. 1, pp. 41-49, 2002.
[25] N. Fazeli, H. R. Marnani, S. Sanjani, R. Zare, S. Dehghan, and N. Jahani, “Seasonal Variation of Copepoda in Chabahar Bay-Gulf of Oman,” Jordan Journal of Biological Sciences, vol. 3, no. 4, pp. 153-164, 2010.
[26] W. Zhang et al., “Onshore–offshore variations of copepod community in northern South China Sea,” Hydrobiologia, vol. 636, no. 1, pp. 257-269, 2009.
[27] R. Kobbi-Rebai, N. Annabi-Trabelsi, H. Khemakhem, H. Ayadi, and L. Aleya, “Impacts of restoration of an uncontrolled phosphogypsum dumpsite on the seasonal distribution of abiotic variables, phytoplankton, copepods, and ciliates in a man-made solar saltern,” Environmental Monitoring and Assessment, vol. 185, no. 3, pp. 2139-2155, 2013.
[28] D. V. Subbamma, “Plankton of a temple pond near Machili Patnam, Andhra Pradesh,” Journal of Aquatic Biology, vol. 7, no. 1, pp. 17-21, 1992.
[29] C. García-Comas et al., “Zooplankton long-term changes in the NW Mediterranean Sea: Decadal periodicity forced by winter hydrographic conditions related to large-scale atmospheric changes?,” Journal of Marine Systems, vol. 87, no. 3, pp. 216-226, 2011.
[30] S. Maiphae and P. Sa-ardrit, “Marine copepods at Mo Ko Thale Tai, Gulf of Thailand,” Songklanakarin Journal of Science and Technology, vol. 33, no. 6, pp. 641-651, 2011.