Muhammad Faisal Wasim1,*, Jawad Ahmad2
1Govt. Dyal Singh Graduate College, Lahore
2University of Engineering and Technology, Lahore-RCET
Received: 10-Aug-2022 / Revised and Accepted: 29-Sep-2022 / Published On-Line: 06-Oct-2022
Abstract:
Nanocomposite has found application in the modern field of technology based on its specific properties. A nanocomposite of alumina and zirconia (Al2O3-ZrO2) was synthesized using a simple Sol-Gel technique and sintered at different temperatures from 500 ºC to 1100 C with an interval of 200 ºC. A thin film was deposited on a 1 mm thick aluminum sheet. The structural simulation of the deposited thin film was analyzed using ANSYS. Particle size and phase are verified by XRD. DSC-TGA (SDT) analysis was used to determine the 1100 ºC of Al2O3/ZrO2 nanocomposite for thin film by dipping method. SEM Material morphology of the synthesized material performed by the SEM method in particle size 90-100nm.
Keywords: Ceramic, thermal resistive, nanocomposite.
Introduction:
Heat transportation through an insulating coating is the demanding aspect in the modern technologies. Heat resistive thinnest thin film or coating is the demand of the industry and scientific instruments. For this purposed different techniques has used such as the insulating spaces, air gabs, sinking metallic setups and fans are used. Weight and thinness has a important aspect of such a issues. Many materials have used forth this purpose but the increasing demand of the ceramics materials of such types of application with significant properties. The Ceramic properties attaining material Zirconium-toughened alumina (ZTA) have main properties of the insulating behaviors. The high hard, polished and smooth surface is also its suitability for coating of the materials as the external coating. The resistance against its wear and tear enhances its application in this regards[1].
The synthesized nanocomposite materials AZO have high thermals expansion such as the Al2O3, having approximate same thermal coefficient of the similar values. The other significant property has its effect for its industrial used such as the thermal insulating with low values of the thermal conductivity in the range of 2-3w/mK. High reducibility of the cracks propagation with values of the 6 to 8 MP (atmosphere) was the important aspect as coating. High strength with ha suitable cohesive ability makes its used effective as the thin coating. [2, 3] Different methodology has used for the synthesis of the materials, such as the hydrothermal [2], wet chemical [3] and chemical bath method [4]. The adopted technique was the Sol-Gel techniques[4].
In this work synthesized stale thermal AZT material has used as a thin film over the aluminum sheet of the thickness 0.5mm with coating of 0.1mm. The dip coating technique has used for deposition of colloidal sticking solution of AZO[5]. Standard digital thermometer with measuring and standard setup has used as shown in the figure. All the setup thin film coated sheet two thermometers fitted in such a way one had in front of the heater and other on the opposite sides of the specimen. A digital heater with controlled flow rate has used for heating purpose from 0-1000ºC with a constant flow rates 1 ºC/mim. The solution 20% molar composition content and 40% molar composition ZrO2 content, affecting the powder size and thus its size-dependent properties as well as agglomeration and surface area. In this work, composite nano-powders Al2O3–20 wt were synthesized by the sol–gel method. % ZrO2 (2 mol% YCl2)[6]. A comparison of the thermometer reading gives a idea about the heat resistive behavior of the coated material.
Experimental procedure and methodology
This experimental procedure consist of three steps, first one is Synthesis ma ZTA nanocomposite [7, 8], second deposition of thin film[9] and third one Heat transportation Analysis.
In the first step, the nanocomposite material has synthesized same [10, 11] as the reference as describes in the schematic shown below in the Fig. 1.

Fig. 1 Experimental setup of material synthesis
In the second step, after the selection of the sample 1100°C, dip coating technique has used for thin deposition on the high graded Alluminium substrate of 1mm. Dip coating technique and procedure has adopted by the reference paper. The colloidal solution of the synthesis material has formed in di-methyle alcohol and dudican oil added for sticking behavior [12, 13].
In third step, we used a crucible material tube in which we arrange the experimental setup consist of two digital thermometer one thermometer act as the reference(A) and the second act as the observed thermometer(B). A heater is used from one side which gives the heat [14].On the other side thermometer note the temperature, the difference between reading the temperatures gives the heat transportation effect from the coated thin film and without coating. By the comparison of the data we observed the behavior of the coated thin film over the substrate [15, 16]. The detail of the thermal behavior analysis explained in Fig.2

Fig. 2 Heat transportation study setup though coated Alluminium sheet
Results and Discussion
3.1 XRD analysis:
The Fig. 3 gives the structural approaching aspect and analysis of the synthesized materials. The peaks behaviors and the crystallographic constants and indexes also have very close agreement with the reference pattern no, “Aluminum Zirconium oxide-00-054-00294”. This graph peaks, broadening and sequences identify the structure and grains size of the synthesized material [17, 18]. The highest temperature synthesis materials have highest clear peaks with same khl values. The graph shows the clear verification of the crystallographic behavior with nanocomposite composition and peaks position[19]. The intensity of these phases increases up to 1100°C.

Fig. 3 Morphological aspect and analysis graph of XRD
1.2 Thermal Analysis
Fig. 4 shows TGA/DSC analysis of the thin film used as coating over the sheet of the aluminum and heat transportation analysis. The analysis has focused here to check and idea about the residual amount and reduction of the organic content at different temperatures. The graphic analysis shows that the synthesized material has no phase change over the 600°C up to the 1000ºC. This analysis help to select the sample sintered at the 1100°C for the use of the coating as the heat resistive layers with an efficient and effective hear resistive behaviors.
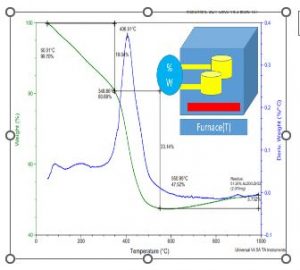
Fig. 4 TGA/DSC Analysis of the thin film used as coating and heat transportation analysis
Fig. 5 graph shows the thermo gravimetric analysis of the two samples sintered at the 1100°C and other was the unsintered material. The change in the area under the curves and removal of the hump of the curves gives a clears idea about it. It’s a three parametric graph of the heat flow, weight loss and the temperatures changes [20, 21].
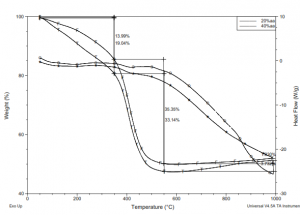
Figure 5 TGA analysis of the coating material
Fig.6 shows the graph between the transportation of the heat and the temperature changes when the experimentation of the heat transportation performed with and without the coating. This shows that a significant heat flow reduction after the coating of the synthesized materials. An effective decrease of the heat transportation was found to be 60%.
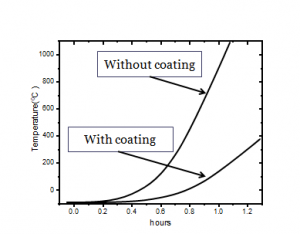
Fig. 6 Coating and without coating temperature increase effect with respect to time
3.2 Optical and Morphological analysis
Fig. 7 shows homogeneous fabrication of the AZO material for the heat resistive ceramics coating. The nanocomposite texture has observed and the particle size was in the range of the 50-100nm. The agglomeration natures observed that an indication of the composite of the two oxides’ materials with great ceramics properties. Fig.8 shows that the optical bandage of the material was the 4.1eV [22, 23].

Fig.7 SEM image soft synthesized of heat resistive coating material
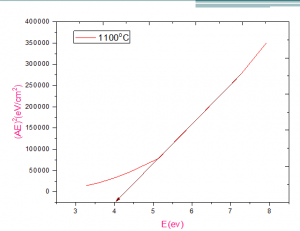
Fig.8 optical band gap analysis of the material
1.2 Structral similation and Heat flow analysy usin ANSYSIS:
The thermal diffusivity of water is much lower on aluminum; heat propagation in water is therefore slower from aluminum. Conduction in stagnant water is simulated for up to 1000 sec. The temperature contours and temperature profiles on line in different time cases are shown in Figures .The stractural and Heat flow simulation was performed by ANSYS software as shown in the figure . In mechanics, equivalent stress is a type of stress that is equal to the same magnitude of elastic stress[24]. The equivalent tensile stress can be calculated from the results of basic uni-axial tensile tests and used to predict the yielding of materials under loading. Heat distribution and heat flow are two tests that make up the heat transfer characteristics study procedure through ANSYS.

Fig. 9 Simulation analysis of the heat flow through the coated thin film over a Alluminium sheet.(A) Structural side view, (B) simulation of side, (C) Structural front view, (D) simulation of side with a controvert vector diagram.
Equivalent Heat flow for ZAT thin film growth aluminum was 0-1000°C. The simulation of the thin film distributes the heat flow into different bands and ranges. With maximum band of red color in range 888 to 1000°C other distribution as shown in the figure above.
Conclusion
Heat resistive coating of the synthesis material was analysis with digital thermometer relative measurement method. This analysis gives the heat resistive ability increase up to 50-60% analysis by the special design crucible heating setup. The coating material morphology found nano-range of 50-100nm with an optical band gape 4.1eV. Simulation result has very close agreement with the experiment bench marks values.
Author’s Contribution: M. F. Wasim, Conceived the idea, Designed the simulated work and data analysis. Jawad Ahmad, help in analysis, write uo and interpretation of data.
Funding: The publication of this article was funded by no one.
Conflicts of Interest: The authors declare no conflict of interest.
REFERENCES
[1] S. Sarker, H. T. Mumu, M. Al-Amin, M. Zahangir Alam, and M. A. Gafur, “Impacts of inclusion of additives on physical, microstructural, and mechanical properties of Alumina and Zirconia toughened alumina (ZTA) ceramic composite: A review,” Materials Today: Proceedings, vol. 62, pp. 2892-2918, 2022/01/01/ 2022.
[2] J. Gao, T. Li, Z. Yan, S. Liu, Y. Zhao, and W. Tong, “Research on the interface and properties of spherical ZTA particles reinforced Fe-Cr-B matrix composite,” Journal of Materials Research and Technology, vol. 19, pp. 1322-1331, 2022/07/01/ 2022.
[3] S. Li, L. Niu, Y. Zhu, J. Chai, Z. Wang, and E. Xie, “Mechanical and thermal properties of ZrC/ZTA composites prepared by spark plasma sintering,” Ceramics International, vol. 48, pp. 6453-6460, 2022/03/01/ 2022.
[4] M. A. J. Selvam, S. Arunkumar, V. Balambica, S. Padmanabhan, T. V. Kumar, and V. S. Shai Sundaram, “Influence of copper anodization on structural and heat transfer characteristic of aluminum piston,” Materials Today: Proceedings, vol. 59, pp. 1407-1413, 2022/01/01/ 2022.
[5] B. A. Witika, V. J. Smith, and R. B. Walker, “Top-Down Synthesis of a Lamivudine-Zidovudine Nano Co-Crystal,” Crystals, vol. 11, p. 33, 2021.
[6] M. B. Gawande, A. Goswami, F.-X. Felpin, T. Asefa, X. Huang, R. Silva, X. Zou, R. Zboril, and R. S. Varma, “Cu and Cu-Based Nanoparticles: Synthesis and Applications in Catalysis,” Chemical Reviews, vol. 116, pp. 3722-3811, 2016/03/23 2016.
[7] M. W. Alam, H. S. Al Qahtani, B. Souayeh, W. Ahmed, H. Albalawi, M. Farhan, A. Abuzir, and S. Naeem, “Novel Copper-Zinc-Manganese Ternary Metal Oxide Nanocomposite as Heterogeneous Catalyst for Glucose Sensor and Antibacterial Activity,” Antioxidants, vol. 11, p. 1064, 2022.
[8] S. Paknia, Z. Izadi, M. Moosaipour, S. Moradi, B. Khalilzadeh, M. Jaymand, and H. Samadian, “Fabrication and characterization of electroconductive/osteoconductive hydrogel nanocomposite based on poly(dopamine-co-aniline) containing calcium phosphate nanoparticles,” Journal of Molecular Liquids, vol. 362, p. 119701, 2022/09/15/ 2022.
[9] W. Zhou, P. Wu, L. Zhang, S. Yao, D. Zhu, and Y. Cai, “Layer-by-layer assembly of nanocomposite interlayers on a kaolin substrate for enhancing membrane performance of Pb(II) and Cd(II) removal,” Science of The Total Environment, vol. 820, p. 153149, 2022/05/10/ 2022.
[10] Y. Song, F. Yang, M. Ma, Y. Kang, A. Hui, Z. Quan, and A. Wang, “Green synthesized Se–ZnO/attapulgite nanocomposites using Aloe vera leaf extract: Characterization, antibacterial and antioxidant activities,” LWT, vol. 165, p. 113762, 2022/08/01/ 2022.
[11] S. Akbarzadeh, L. Sopchenski Santos, V. Vitry, Y. Paint, and M.-G. Olivier, “Improvement of the corrosion performance of AA2024 alloy by a duplex PEO/clay modified sol-gel nanocomposite coating,” Surface and Coatings Technology, vol. 434, p. 128168, 2022/03/25/ 2022.
[12] K. S. Burts, T. V. Plisko, V. G. Prozorovich, G. B. Melnikova, A. I. Ivanets, and A. V. Bildyukevich, “Development and Study of PVA–SiO2/poly(AN-co-MA) Dynamic Nanocomposite Membranes for Ethanol Dehydration via Pervaporation,” Membranes and Membrane Technologies, vol. 4, pp. 101-110, 2022/04/01 2022.
[13] L. Sarango, L. Paseta, M. Navarro, B. Zornoza, and J. Coronas, “Controlled deposition of MOFs by dip-coating in thin film nanocomposite membranes for organic solvent nanofiltration,” Journal of Industrial and Engineering Chemistry, vol. 59, pp. 8-16, 2018/03/25/ 2018.
[14] X. Chen, “Synthesis and characterization of ATO/SiO2 nanocomposite coating obtained by sol–gel method,” Materials Letters, vol. 59, pp. 1239-1242, 2005/04/01/ 2005.
[15] O. Razavizadeh and M. Ghorbani, “Sol–gel preparation and corrosion properties of Zn–SiO2 nanocomposite thin film,” Journal of Sol-Gel Science and Technology, vol. 79, pp. 133-143, 2016/07/01 2016.
[16] A. A. Silva, R. A. Pinheiro, C. d. A. Razzino, V. J. Trava-Airoldi, and E. J. Corat, “Thin-film nanocomposites of BDD/CNT deposited on carbon fiber,” Diamond and Related Materials, vol. 75, pp. 116-122, 2017/05/01/ 2017.
[17] H. Zaka, S. S. Fouad, B. Parditka, A. E. Bekheet, H. E. Atyia, M. Medhat, and Z. Erdélyi, “Enhancement of dispersion optical parameters of Al2O3/ZnO thin films fabricated by ALD,” Solar Energy, vol. 205, pp. 79-87, 2020/07/15/ 2020.
[18] D. Gupta, V. Chauhan, N. Koratkar, F. Singh, A. Kumar, S. Kumar, and R. Kumar, “High energy (MeV) ion beam induced modifications in Al2O3-ZnO multilayers thin films grown by ALD and enhancement in photoluminescence, optical and structural properties,” Vacuum, vol. 192, p. 110435, 2021/10/01/ 2021.
[19] A. A. Chaaya, R. Viter, I. Baleviciute, M. Bechelany, A. Ramanavicius, Z. Gertnere, D. Erts, V. Smyntyna, and P. Miele, “Tuning Optical Properties of Al2O3/ZnO Nanolaminates Synthesized by Atomic Layer Deposition,” The Journal of Physical Chemistry C, vol. 118, pp. 3811-3819, 2014/02/20 2014.
[20] J. Cortez-Elizalde, A. A. Silahua-Pavón, G. E. Córdova-Pérez, J. C. Arévalo-Pérez, Z. Guerra-Que, C. G. Espinosa-González, F. Ortíz-Chi, S. Godavarthi, and J. G. Torres-Torres, “Production of 5-Hydroxymethylfurfural from glucose using Al2O3-TiO2-ZrO2 ternary catalysts,” Catalysis Today, vol. 392-393, pp. 116-130, 2022/06/01/ 2022.
[21] Y. Bai, S. W. Lee, H. Chen, F. Li Yu, and J. F. Yang, “Characterization of the Near-Eutectic Al2O3–40 wt% ZrO2 Composite Coating Fabricated by Atmospheric Plasma Spray. Part I: Preparation and Characterization of Nano Al2O3–ZrO2 Powder Using Sol–Gel Method,” Materials and Manufacturing Processes, vol. 26, pp. 330-337, 2011/01/31 2011.
[22] J. I. Peña, R. I. Merino, N. R. Harlan, A. Larrea, G. F. de la Fuente, and V. M. Orera, “Microstructure of Y2O3 doped Al2O3–ZrO2 eutectics grown by the laser floating zone method,” Journal of the European Ceramic Society, vol. 22, pp. 2595-2602, 2002/01/01/ 2002.
[23] J. Gao, W. Su, X. Song, Y. Bai, J. Wang, and G. Yu, “Post-mortem analysis of Cr2O3-Al2O3-ZrO2 refractory bricks used in an industrial opposed multi-burner gasifier,” Engineering Failure Analysis, vol. 134, p. 106017, 2022/04/01/ 2022.
[24] E. Postek and T. Sadowski, “Impact model of the Al2O3/ZrO2 composite by peridynamics,” Composite Structures, vol. 271, p. 114071, 2021/09/01/ 2021.